Price 61288.0 आईएनआर/ Box
MOQ : 1 Box
Specification
- संकेत
- Cancer treatment
- उत्पत्ति
- India
- नमक की संरचना
- Trastuzumab
- खुराक प्रपत्र
- Injection
- स्टोरेज निर्देश
- Cool and dry place
- शेल्फ लाइफ
- महीने
Trade Information
- Minimum Order Quantity
- 1 Box
- आपूर्ति की क्षमता
- प्रति महीने
- डिलीवरी का समय
- दिन
About
Canmab Trastuzumab 440 Mg Injection stands as a sterling, meritorious choice in oncological care, earning ineffable trust through favorable reviews from the medical community. Formulated with the powerful salt composition Trastuzumab, this injection is indicated for cancer treatment, particularly breast cancer. Manufactured in India and exported with rigorous quality oversight, Canmab offers exceptional value and reliability. For optimal efficacy, store in a cool, dry place. With a shelf life of 12 months, Order Today to experience the benefits trusted by healthcare professionals worldwide.
Key Applications and Usage Areas
Canmab Trastuzumab 440 Mg Injection is widely applied in oncology clinics, specialized hospitals, and cancer treatment centers. Its principal area of application is in the management of HER2-positive breast cancer, administered under the supervision of qualified medical professionals. The usage type is strictly by intravenous injection, tailored to patient-specific protocols. Canmab's proven effectiveness in targeted cancer therapy sets it apart as a valuable component in contemporary cancer care.
Sample Policy, Payment Terms, and Delivery Process
For Canmab Trastuzumab 440 Mg Injection, sample requests are handled with a practical approach to meet client preferences within the domestic market. Payment terms are designed with transparency at the core, securing access to the lowest price offers. Shipped goods are dispatched promptly, with typical delivery times ranging from a few days to a week depending on order location within India. Reliability in both products and shipment is of paramount importance.
Key Applications and Usage Areas
Canmab Trastuzumab 440 Mg Injection is widely applied in oncology clinics, specialized hospitals, and cancer treatment centers. Its principal area of application is in the management of HER2-positive breast cancer, administered under the supervision of qualified medical professionals. The usage type is strictly by intravenous injection, tailored to patient-specific protocols. Canmab's proven effectiveness in targeted cancer therapy sets it apart as a valuable component in contemporary cancer care.
Sample Policy, Payment Terms, and Delivery Process
For Canmab Trastuzumab 440 Mg Injection, sample requests are handled with a practical approach to meet client preferences within the domestic market. Payment terms are designed with transparency at the core, securing access to the lowest price offers. Shipped goods are dispatched promptly, with typical delivery times ranging from a few days to a week depending on order location within India. Reliability in both products and shipment is of paramount importance.
FAQ's of Canmab Trastuzumab 440 Mg Injection:
Q: How should Canmab Trastuzumab 440 Mg Injection be stored for best efficacy?
A: Canmab Trastuzumab 440 Mg Injection should be stored in a cool and dry place away from direct sunlight to maintain its stability and effectiveness.Q: What is the main indication for using Canmab Trastuzumab 440 Mg Injection?
A: This injection is primarily indicated for the treatment of HER2-positive cancers, most commonly breast cancer, as part of a supervised oncology regimen.Q: When can I expect the shipment after placing my order for Canmab Trastuzumab 440 Mg Injection?
A: Orders are generally processed promptly, with shipped goods delivered within a few days to one week, depending on your location within India.Q: Where is Canmab Trastuzumab 440 Mg Injection manufactured?
A: Canmab Trastuzumab 440 Mg Injection is manufactured and exported from India, adhering to stringent quality standards.Q: What is the process for ordering samples of Canmab Trastuzumab 440 Mg Injection?
A: Clients may request samples based on the outlined sample policy, typically by contacting the supplier directly to discuss availability and sample arrangements.Q: How is Canmab Trastuzumab 440 Mg Injection administered?
A: It is administered by intravenous injection and should always be given under the supervision of a healthcare professional.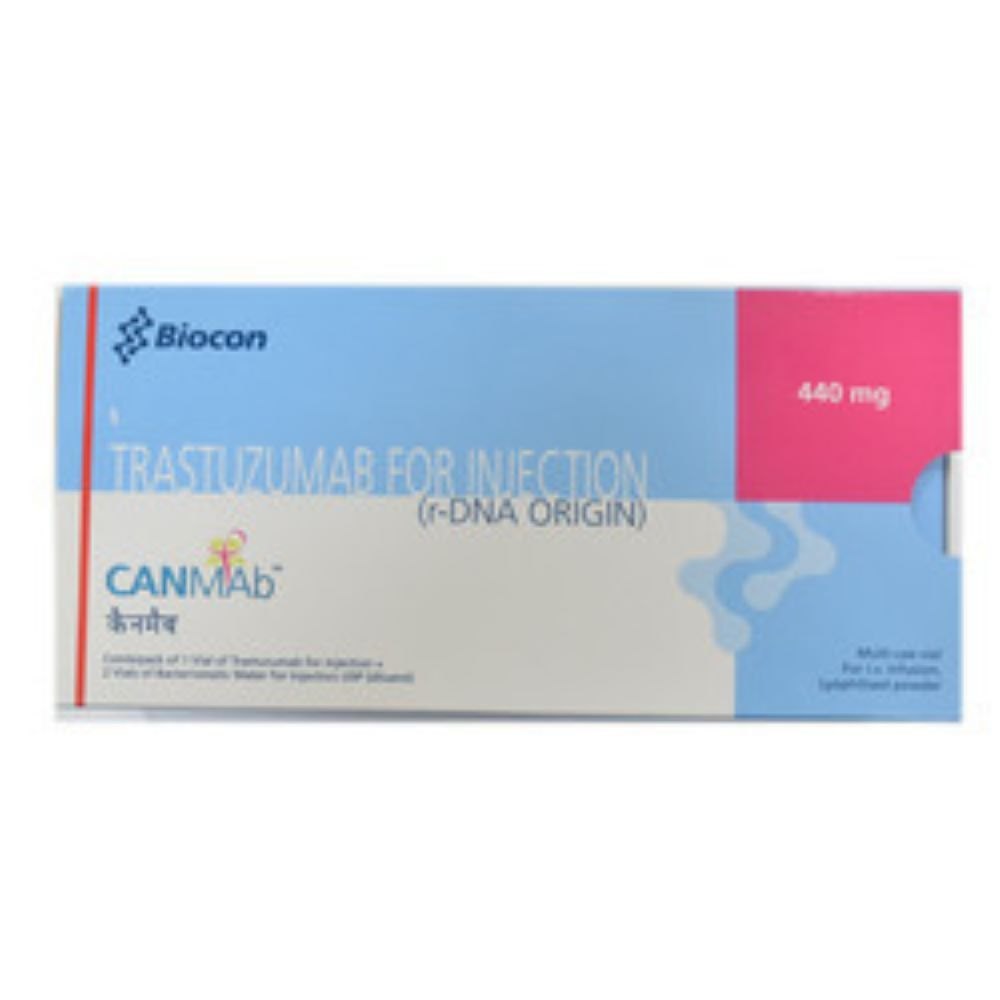

Tell us about your requirement

Price: Â
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
मोबाइल number
Email

 जांच भेजें
जांच भेजें जांच भेजें
जांच भेजें