Price 58820.0 आईएनआर/ Bottle
MOQ : 1 Bottle
Specification
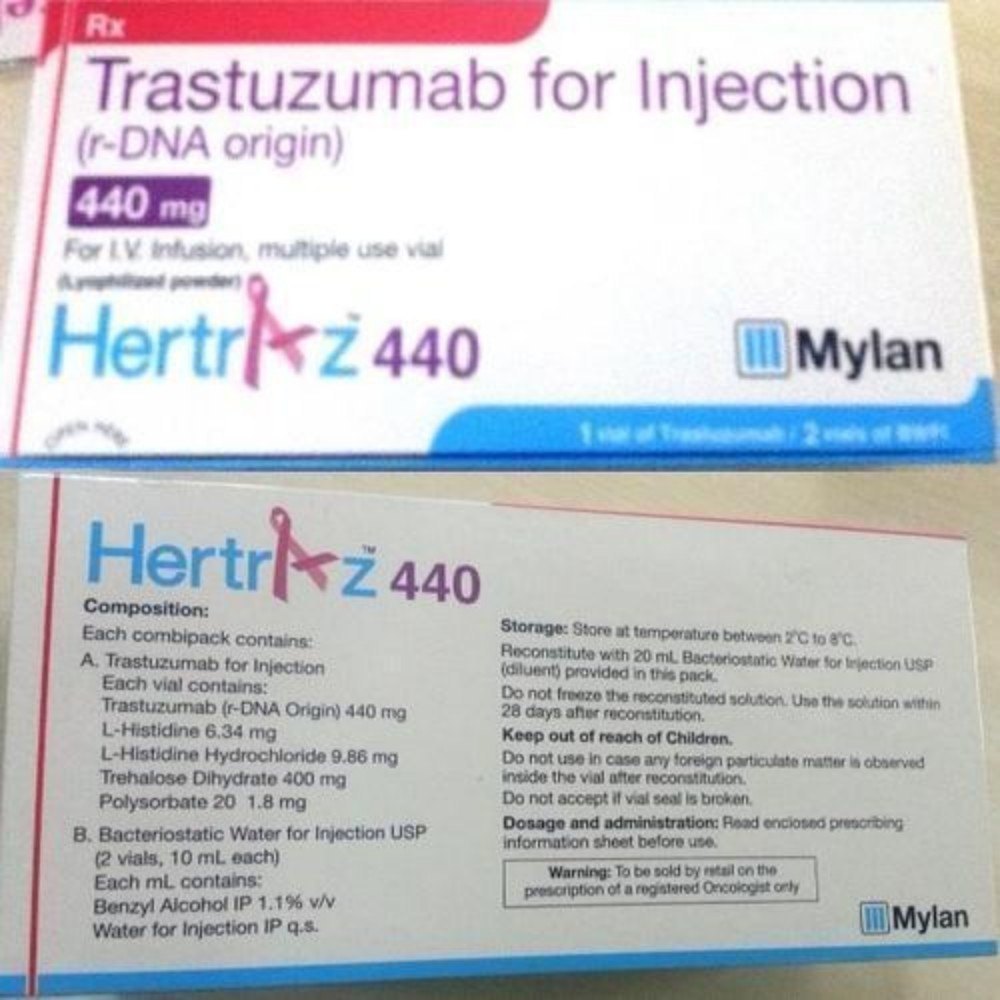
- नमक की संरचना
- Trastuzumab
- उत्पत्ति
- India
- खुराक प्रपत्र
- Injection
- संकेत
- Cancer treatment
- स्टोरेज निर्देश
- Cool and dry place
- शेल्फ लाइफ
- महीने
Trade Information
- Minimum Order Quantity
- 1 Bottle
- आपूर्ति की क्षमता
- प्रति महीने
- डिलीवरी का समय
- दिन
About
Add to Cart the featured Hertraz Trastuzumab 440 Mg Injection-an unrivaled, sought-after solution for cancer treatment. Manufactured in India and exported globally, this world-class oncology product contains a matchless composition of Trastuzumab, meticulously formulated for maximum therapeutic benefit. The top product in its category, Hertraz offers a shelf life of 12 months and is supplied by leading exporters and suppliers in India. Dosage form: injection. Store in a cool, dry place to ensure potency and safety for optimal results during treatment.
Key Features & Advanced Applications
Hertraz Trastuzumab 440 Mg Injection boasts top-tier, world-class features engineered for cancer therapy. Its matchless composition is designed for precision in oncology applications, making it highly sought-after in various healthcare facilities. Other notable attributes include its reliable shelf life and ease of use. Plant applications are focused on pharmaceutical manufacturing, ensuring high purity and efficacy. These outstanding features solidify Hertraz's reputation as a leading injectable in cancer care.
Sample Availability & Export Policy
Sample of Hertraz Trastuzumab 440 Mg Injection is available upon request for those in domestic mar and international destinations. We offer competitive rates and diligent support for sample inquiries. Our sample policy is straightforward: connect with us to discuss terms tailored for your requirements. Main export markets include Asia, Africa, and Europe, underscoring our commitment as a top supplier to global healthcare providers seeking quality oncology solutions.
Key Features & Advanced Applications
Hertraz Trastuzumab 440 Mg Injection boasts top-tier, world-class features engineered for cancer therapy. Its matchless composition is designed for precision in oncology applications, making it highly sought-after in various healthcare facilities. Other notable attributes include its reliable shelf life and ease of use. Plant applications are focused on pharmaceutical manufacturing, ensuring high purity and efficacy. These outstanding features solidify Hertraz's reputation as a leading injectable in cancer care.
Sample Availability & Export Policy
Sample of Hertraz Trastuzumab 440 Mg Injection is available upon request for those in domestic mar and international destinations. We offer competitive rates and diligent support for sample inquiries. Our sample policy is straightforward: connect with us to discuss terms tailored for your requirements. Main export markets include Asia, Africa, and Europe, underscoring our commitment as a top supplier to global healthcare providers seeking quality oncology solutions.
FAQ's of Hertraz Trastuzumab 440 Mg Injection:
Q: How should Hertraz Trastuzumab 440 Mg Injection be stored to ensure efficacy?
A: Hertraz Trastuzumab 440 Mg Injection must be stored in a cool, dry place to maintain its stability and effectiveness. Always check the storage instructions provided on the packaging.Q: What is the main usage of Hertraz Trastuzumab 440 Mg Injection?
A: The main indication for Hertraz Trastuzumab 440 Mg Injection is the treatment of certain types of cancers, most notably HER2-positive breast cancer. It's administered as an injection under medical supervision.Q: Where is Hertraz Trastuzumab 440 Mg Injection manufactured and supplied from?
A: Hertraz Trastuzumab 440 Mg Injection is manufactured in India and supplied by reliable exporters and suppliers across the globe, making it accessible in various regions including Asia, Africa, and Europe.Q: What is the shelf life of Hertraz Trastuzumab 440 Mg Injection?
A: The shelf life of Hertraz Trastuzumab 440 Mg Injection is 12 months from the date of manufacture. Always verify the expiration date before use.Q: How can a sample of Hertraz Trastuzumab 440 Mg Injection be obtained?
A: Samples are available upon request. Contact the supplier directly to discuss an offer that matches your requirements and market demands. Sample policy terms will be shared with interested buyers.
Tell us about your requirement

Price: Â
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
मोबाइल number
Email
अधिक Products in कैंसर रोधी औषधियाँ Category
300 मिलीग्राम टेनोफोविर डिसोप्रोक्सिल फ्यूमरेट टैबलेट आईपी
माप की इकाई : पैक/पैक
स्टोरेज निर्देश : कमरे का तापमान।
न्यूनतम आदेश मात्रा : 100
मूल्य की इकाई : पैक/पैक
मूल्य या मूल्य सीमा : आईएनआर

 जांच भेजें
जांच भेजें जांच भेजें
जांच भेजें