- Home Page
- Company Profile
-
Our Products
- Brain Nervous And Anti Depresant Medicines
- 25 MG Atom-oxetine Hydrochloride Tablets
- 40 MG Atomo-xetine Hydrochloride Tablets
- 150 MG Bupropion Hydrochloride Tablets IP
- Pharma 30 MG Mirtaza-pine Tablets IP
- 30 MG Mirtaza-pine Tablets IP
- Piracetam And Citicoline Tablets
- Pregabalin Injection
- Neoprate Sodium Valproate Injection 100mg
- Pircetam Piracetam Tablet 400mg
- Steroid Medicines
- 5 GM Pharma Gel
- 100 MG pharma Decanoate Injection IP
- 250 MG pharma Injection
- 250 MG pharma Cypionate Injection
- pahrma Gel 1% W-W
- Headon Somatropin Injection 4IU
- Isotroy Isoflurane 100 ml
- Nacphin Nalbuphine Injection 10 mg
- Neo-Drol Methylprednisolone Injection 1000mg
- Premaxo Methylprednisolone Injection 80mg
- Ropin Ropivacaine Injection 0.2%
- Troyhep Heparin Injection 25000IU
- Sustanon Testosterone 250 Mg Injection.
- Zydip Beclometasone Lotion 100ml
- Anti Cancer Medicines
- 800 MG Piracetam Tablets
- 25 MG Tenofovir Alafenamide Tablets
- 440 MG Trastuzumab For Injection
- 5 MG Minoxidil Tablets IP
- 1.5 MG Levonorgestrel Tablets IP
- 1200 MG Piracetam Tablets
- 300 MG Tenofovir Disoproxil Fumarate Tablets IP
- 440 MG Trastuzumab Lyophilized Powder For Concentrate For Solution For Infusion
- Abemy Bevacizumab Injection 400mg
- Adriamycin Doxorubicin HCL Injection
- Adriamycin Doxorubicin HCL 10ml Injection
- Adrim Doxorubicin HCL 50mg/25ml Injection
- Epofit Erythropoietin alfa 2000 IU
- Epofit Recombinant Human Erythropoietin Alfa Injection 4000 IU
- Epofit Recombinant Human Erythropoietin Alfa Injection 5000 IU
- Abirapro Abiraterone Acetate Tablet 250mg
- AltrAZ Anastrozole Tablet 1 mg
- Alrubicin Epirubicin Injection 100 Mg
- Anabrez Anastrozole Tablet IP 1 MG
- Anaday Anastrozole Tablet 1mg
- Aprecap Aprepitant 125/80 Capsule
- Aprecap Fosaprepitant Injection 150mg
- Apretero Aprepitant 125/80 Mg Kit
- Arimidex Anastrozole Tablet 1mg
- Armotraz Anastrozole Tablet 1mg
- Bevacirel Injection 100 Mg
- Bevacirel Bevacizumab Injection 400Mg
- Bevatas Bevacizumab Injection 100 Mg
- Bevatas Bevacizumab 400
- Bimode Injection 100mg
- Biocure Bortezomib Injection 2mg
- Biotrexate Tablet 2.5mg
- Biotrexate Methotrexate Injection 15mg
- Biotrexate Methotrexate Injection 50 MG
- Biotrexate Methotrexate Injection 500 mg
- Bortecad Bortezomib Injection 2mg
- Bortenat Bortezomib Injection 2mg
- Bortenat Bortezomib Injection 3.5 Mg
- Borviz Bortezomib Injection 2.5 Mg
- Borviz Bortezomib Injection 2mg
- Bryxta Bevacizumab Injection 100 mg
- Cacit Capecitabine Tablet 500 Mg
- Capegard Capecitabine Tablet 500 mg
- Capiibine Capecitabine Tablet 500 mg
- Zyceva Erlotinib Tablet 150 mg
- Abevmy Bevacizumab Injection 400 mg
- Adrim Doxorubicin Injection 10 mg
- Alrubicin Epirubicin Injection 50 mg
- Bdron Abiraterone Acetate Tablets 250mg
- Bdron Abiraterone Acetate Tablets 500Mg
- Bendit Bendamustine Injection 100 mg
- Bryxta Bevacizumab Injection 400 mg
- Capnat Capecitabine Tablets 500mg
- Capsy Capecitabine Tablets 500 mg
- Cizumab Bevacizumab 100 mg
- Cizumab Bevacizumab 400 mg
- Cycloxan Cyclophosphamide Tablets 50mg
- Cytax Paclitaxel Injection IP 30 mg
- Cytax Paclitaxel Injection 100 Mg
- Cytax Paclitaxel Injection 260 mg
- Cytax Paclitaxel Injection 300 mg
- Cytogem Gemcitabine Injection 200 mg
- Cytoplatin Cisplatin Injection 10 mg
- Cytoplatin Cisplatin Injection 50 mg
- Daxotel Docetaxel Injection 20 mg
- Daxotel Docetaxel Injection 80mg
- Daxotel Docetaxel Injection 120mg
- Docetax Docetaxel INJECTION 20mg
- Docetax Docetaxel Injection 80 mg
- Docetax Docetaxel Injection 120mg
- Docetere Docetaxel Injection 120 mg
- Docetere Docetaxel Injection 80 mg
- Docetere Docetaxel Injection 120mg
- Emgrast Filgrastim Injection 300mcg
- Endoxan Cyclophosphamide Tablet 50mg
- Endoxan Cyclophosphamide Injection 200 mg
- Endoxan N Cyclophosphamide Injection 1gm
- Endoxan N Cyclophosphamide Injection 500 mg
- Enfiera Rituximab Injection 100mg
- Enfiera Rituximab Injection 500 mg
- Epithra Epirubicin Injection 100 Mg
- Epithra Epirubicin Injection 50 Mg
- Epithra Epirubicin Injection 100 Mg
- Eriocip Erlotinib Tablet 100 Mg
- Erlocip Erlotinib Tablet 150 mg
- Erlonat Erlotinib Tablet 100 mg
- Erlonat Erlotinib Tablets 150 mg
- Evermil Everolimus Tablet 5 MG
- Evermil Everolimus Tablet 10 mg
- Evertor Everolimus Tablets 5mg
- evertor Everolimus Tablets 10 Mg
- Farmorubicin Epirubicin Injection 50mg
- Femistra Anastrozole Tablet 1 Mg
- Fempro Letrozole Tablet 2.5 mg
- Geffy Gefitinib Tablet 250mg
- Gefitero Gefitinib Tablet 250 mg
- Geftib Gefitinib Tablets mg
- Gefticip Gefitinib Tablet 250 mg
- Geftinat Gefitinib Tablets 250 mg
- Gemcite Gemcitabine Injection 1Mg
- Gemcite Gemcitabine Injection 200mg
- Gemita GEMCITABINE Injection 1.4 GM
- Gemita Gemcitabine Injection 1 GM
- Gemita Gemcitabine Injection 200 mg
- Gemtaz Gemcitabine Injection 1 mg
- Gemtaz Gemcitabine Injection 200 mg
- Gemtero Gemcitabine Injection 1 mg
- Genexol Paclitaxel Injection 100MG
- Grafeel Filgrastim Injection 300mcg
- Grastim Filgrastim Injection 300mcg
- Holoxan Ifosfamide Injection 1gm
- Holoxan IFOSFAMIDE-2GM + MESNA-100MG Injection
- Imanib Imatinib Tablets 100mg
- Imanib Imatinib Tablet 400mg
- Imat Imatinib Tablet 100mg
- Imat Imatinib Tablet 400 mg
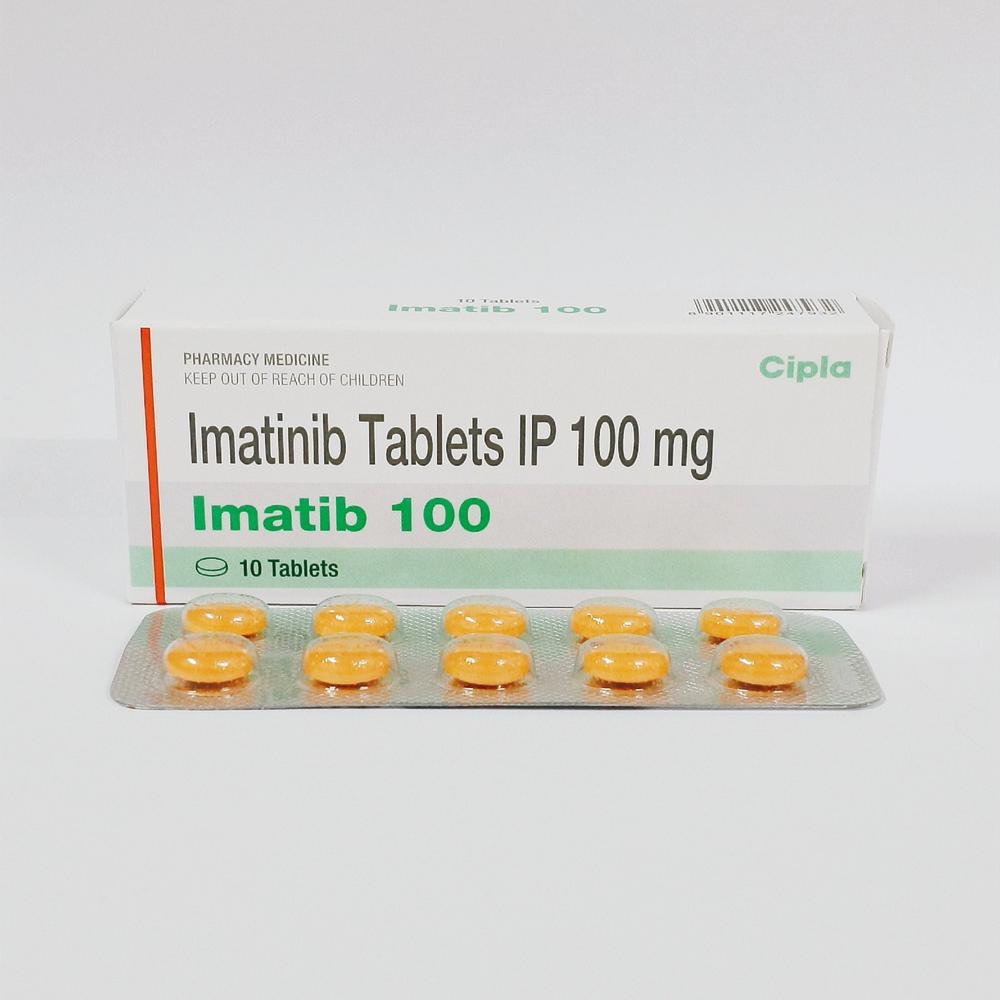
- Imatib Imatinib Tablet 100 Mg
- Imatib Imatinib Tablet 400 mg
- Intaxel Paclitaxel Injection 30 mg
- Intaxel Paclitaxel Injection 100 mg
- Intaxel Paclitaxel Injection 260 Mg
- Irinotel Irinotecan Injection 40 mg
- Irinotel Irinotecan Injection 100 mg
- Irnocam- 40 Irinotecan Injection 20Mg
- Kemocarb Carboplatin Injection 150 mg
- Kemocarb Carboplatin Injection 450 mg
- Lenalid Lenalidomide Capsule 5mg
- Lenalid Lenalidomide Capsule 10 mg
- Lenalid Lenalidomide Capsule 25 mg
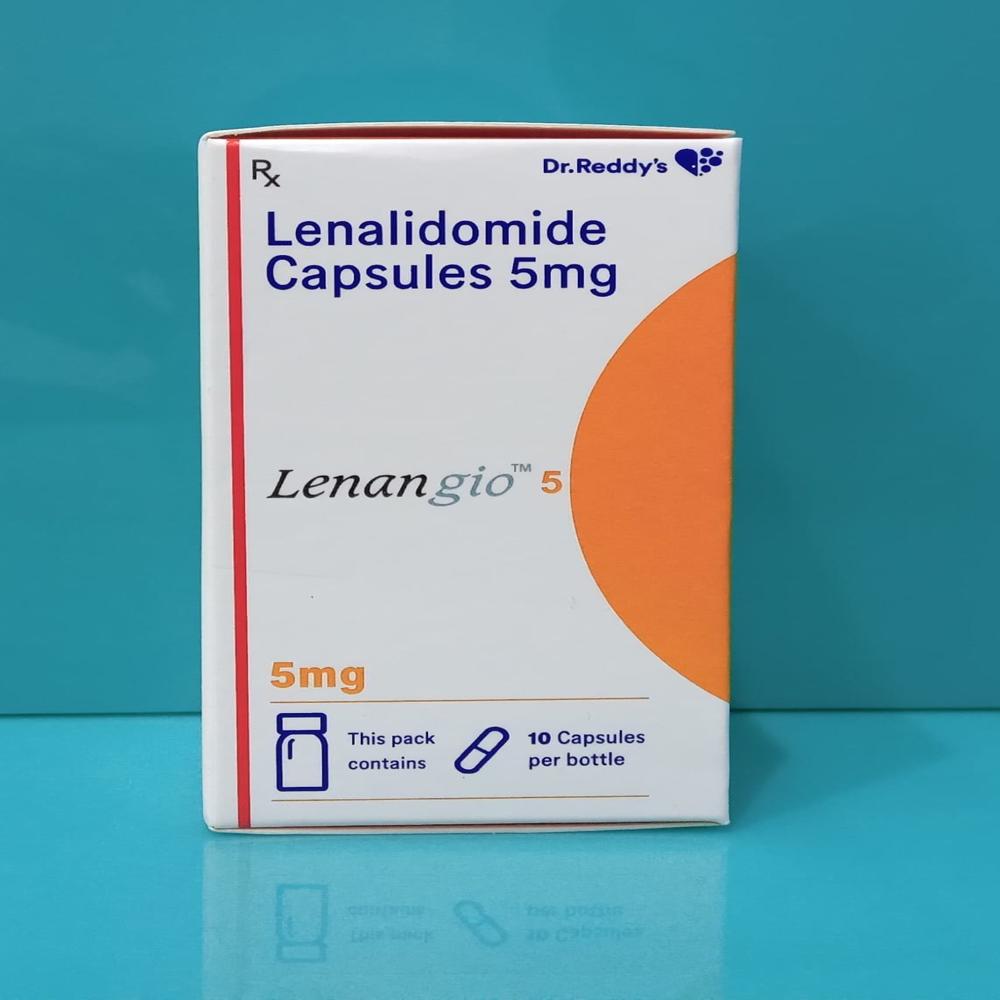
- Lenangio Lenalidomide Capsule 5mg
- Lenangio Lenalidomide Capsule 10mg
- Lenangio Lenalidomide Capsule 25 mg
- Letroz Let-rozole Tablet 2.5 mg
- Lupride Depot Leuprolide Injection 3.75mg
- Lupride Depot Leuprolide Injection 11.25 mg
- Lupride Depot Leuprolide Injection 22.5mg
- Luprodex Depot Leuprolide Injection 3.75mg
- Luprodex Leuprolide Injection 11.25 mg
- Luprodex Leuprolide Injection 22.5 mg
- Maball Rituximab Injection 100 mg
- Maball Rituximab Injection 500mg
- Mabtas Rituximab Injection 100 mg
- Mabtas Rituximab Injection 500mg
- Mabtas RA Rituximab Injection 500mg
- Megaplat Oxaliplatin Injection 50mg
- Megaplat Oxaliplatin Injection 100 mg
- Mitinab Imatinib Tablets 400mg
- Mitotax Paclitaxel Injection 30 mg
- Mitotax Paclitaxel Injection 100 mg
- Mitotax Paclitaxel Injection 250MG
- Mitotax Paclitaxel Injection 300 mg
- Myezom Bortezomib Injection 2mg
- Nanoxel Paclitaxel Injection 30 mg
- Nanoxel Paclitaxel Injection 100MG
- Nanoxel Paclitaxel Injection 300MG
- Neukine Filgrastim Injection 300mcg
- Nufil SFs Injection 300mcg
- Oncolet Let-rozole Tablets 2.5mg
- Oreta Letro-zole Tablets 2.5mg
- Oxaltero Oxaliplatin Injection 50MG
- Oxaltero Oxaliplatin Injection 100mg
- Oxiplat Oxaliplatin Injection 50 mg
- Oxiplat Oxaliplatin Injection 100 mg
- Oxitan Oxaliplatin Injection 50mg
- Oxitan Oxaliplatin Injection 100 mg
- paclitax Paclitaxel Injection 30 mg
- Paclitax Paclitaxel Injection 260mg
- Paclitax Nab Paclitaxel Injection 100mg
- Pegadria Doxorubcin Injection 20MG
- Pegasta Pegfilgrastim Injection 6 mg
- Pegex Pegfilgrastim Injection 6mg
- Pegfeel Granulocyte Colony Stimulating Factor Injection 6 mg
- Pegstim Pegfilgrastim Injection 6mg
- Pemnat Pemetrexed Injection 100mg
- Pemnat Pemetrexed Injection 500mg
- Purplz Bendamustine Injection 100mg
- Reditux Rituximab Injection 100mg
- Reditux Rituximab Injection 500mg
- Religrast Filgrastim Injection 300MCG
- Ristova Rituximab Injection 100mg
- Ristova Rituximab Injection 500mg
- Rituxirel Rituximab Injection 100 mg
- Rituxirel Rituximab Injection 500mg
- Rolimus Everolimus Tablet 5mg
- Rolimus Everolimus Tablet 10 mg
- Samtica Abiraterone Acetate Tablets 250 mg
- samtica Abiraterone Acetate Tablets 500mg
- Sorafenat Sorafenib Tablets 200mg
- Soranib Sorafenib Tablets 200mg
- Stricarb Carboplatin Injection 450 mg
- Taxonab Paclitaxel Injection 100 mg
- Taxotere Docetaxel Injection 20mg
- Taxotere Docetaxel Injection 80mg
- Temoside Temozolomide Capsule 20mg
- Temoside Temozolomide Capsule 100mg
- Temoside Temozolomide Capsule 250 mg
- Veenat Imatinib Capsule 100mg
- Veenat Imatinib Tablets 400mg
- Votrient Pazopanib Tablet 400mg
- Xbira Abiraterone Acetate Tablet 250mg
- X-Plat Oxaliplatin Injection 50mg
- Zecyte Abiraterone Acetate Tablets 250mg
- Zelgor Abiraterone Acetate Tablets 250 mg
- Zoladex Goserelin acetate Injection 3.6 mg
- Zoladex Goserelin acetate Injection 10.8 mg
- Zyceva Erlotinib Tablet 100 mg
- 6-MP Mercaptopurine Tablet 50 mg
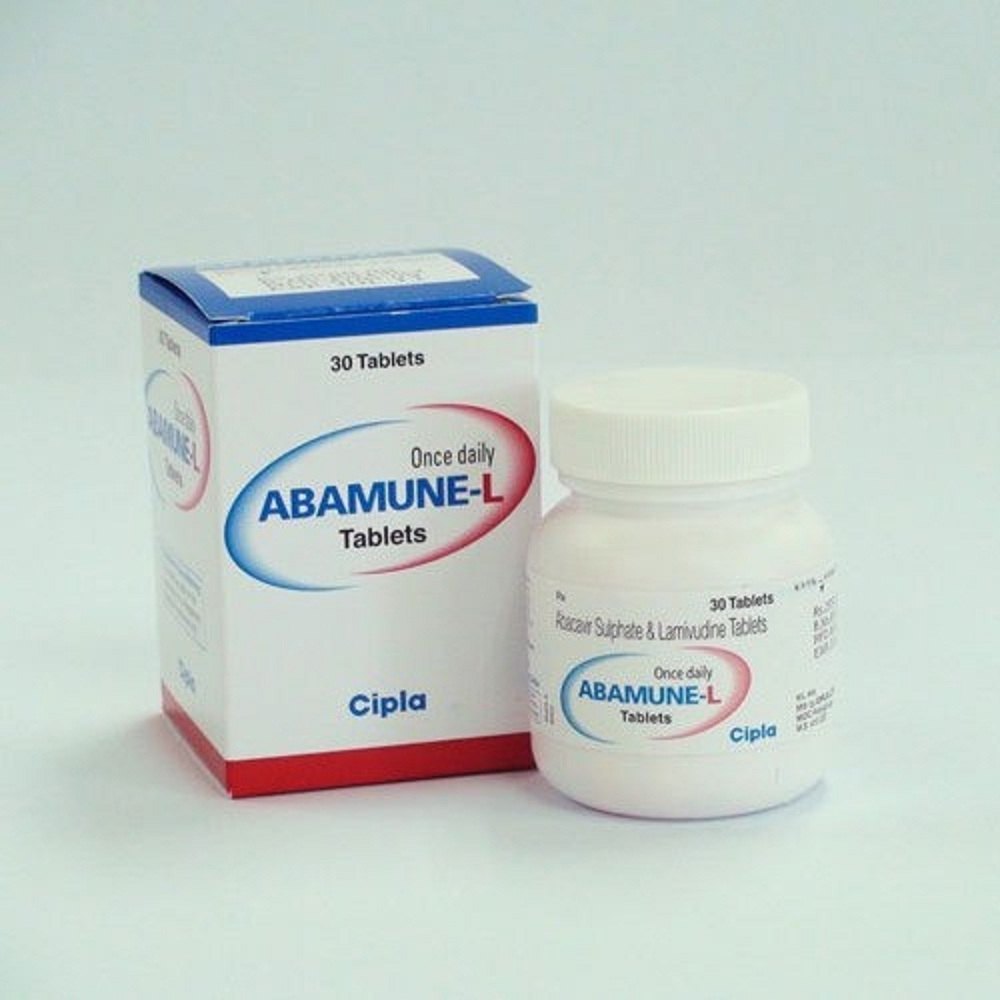
- Abamune L Abacavir & Lamivudine Tablets
- Abamune Abacavir Tablets 300mg
- Abec L Abacavir 600 mg+Lamivudine 300 mg Tablet
- Alfakit Alpha Ketoanalogue Tablets
- Alfalog Alpha Ketoanalogue Tablet
- Arpimune Me Ciclosporin Capsule 25 mg
- Arpimune ME Ciclosporin Capsule 50 mg
- Arpimune ME Ciclosporin Capsule 100mg
- Arpimune ME Oral Solution Ciclosporin 100mg
- Arsenox Arsenic Trioxide Injection 10 mg
- Atazor-R Atazanavir 300mg + Ritonavir 100mg Tablet
- Azadine Azacitidine Injection 100mg
- Azoran Azathioprine Tablet IP 50 mg
- Bdenza Enzalutamide Capsule 40mg
- Bdenza Enzalutamide Capsule 80 mg
- Biomab Nimotuzumab Injection 50mg
- Canmab Trastuzumab Injection 150mg
- Canmab Trastuzumab Injection440 mg
- Celkeran Chlorambucil Tablet 2mg
- Celkeran Chlorambucil Tablet 5mg
- Cyclophil ME Ciclosporin Capsule 25mg
- Cyclophil ME Ciclosporin Capsule 50 mg
- Cyclophil ME Ciclosporin Capsule 100 mg
- Cyclophil ME Oral Solution Ciclosporin 100 mg
- Cytodrox Hydroxyurea Capsule 500 mg
- Dacihep Daclatasvir Tablet 60mg
- Daclahep Daclatasvir Tablet 60mg
- Danavir DARUNAVIR-600MG + RITONAVIR-100MG Tablet
- Daruvir Darunavir Tablet 800mg
- Duovir Lamivudine (150mg) + Zidovudine (300mg) Tablet
- Duovir N Lamivudine (150mg) + Zidovudine (300mg) + Nevirapine (200mg) Tablet
- Efavir Efavirenz Tablet 600 mg
- Endace Megestrol Tablet 160 mg
- Entaliv Entecavir Tablet 0.5 mg
- Entavir Entecavir Tablet 0.5mg
- Entavir Entecavir Tablet 1mg
- Entehep Entecavir Tablet 0.5 mg
- Entehep Entecavir Tablet 1 mg
- Enzamide Enzalutamide Capsule 40 mg
- Erbitux Cetuximab Injection 100mg
- Exemptia Adalimumab Injection 40mg
- Fulvenat Fulvestrant Injection 250
- Gavir Ganciclovir Capsule 250 mg
- Gavir Ganciclovir Injection 500mg
- Glioz Temozolomide Capsule 20 mg
- Glioz Temozolomide Capsule 100 mg
- Glioz Temozolomide Capsule 250 mg
- Hepbest Tenofovir Alafenamide Tablet 25 mg
- Hepcfix Daclatasvir Tablet 60MG
- Hepcinat Sofosbuvir Tablet 400mg
- Hepcinat LP Ledipasvir (90mg) + Sofosbuvir (400mg) Tablet
- Hepcinat LP Tablet Ledipasvir 90mg + Sofosbuvir 400mg Tablet
- Hepcvel Sofosbuvir 400mg & Velpatasvir 100mg Tablet
- Hepcvir Sofosbuvir Tablet 400 mg
- Herclon Trastuzumab Injection 440 mg
- Herduo Lapatinib Tablet 250 mg
- Hermab Trastuzumab Injection 440mg
- Hertraz Trastuzumab Injection 150 mg
- Hertraz Trastuzumab Injection 440mg
- Honvan Fosfestrol Tablet 120 mg
- Hydrosar Hydroxyurea Capsule 500 mg
- Instgra Dolutegravir Tablet 50 mg
- Kabanat Cabazitaxel Injection 60mg
- Kabanat Cabazitaxel Injection 60mg
- Ketoalfa Alpha Ketoanalogue Tablet 200 mg
- Ketograce Alpha Ketoanalogue Tablet
- Ketosteril Alpha Ketoanalogue Tablet
- Lamivir S Lamivudine 150mg + Stavudine 40mg Tablet
- Lazid N Lamivudine 150mg & Zidovudine 300mg+ Nevirapine 200mg
- Ledifos Ledipasvir & sofosbuvir Tablet
- Ledihep Ledipasvir 90mg & sofosbuvir 400mg
- Lopimune Ritonavir (50mg) + Lopinavir (200mg) Tablet
- Myaza Azacitidine Injection 100mg
- Mydekla Daclatasvir Tablet 60 mg
- MY Hep Sofosbuvir Tablet 400mg
- Myhep ALL Sofosbuvir 400mg + Velpatasvir 100mg Tablet
- Myhep Lvir Ledipasvir (90mg) + Sofosbuvir (400mg) Tablet
- Natclovir Ganciclovir Capsule 250 MG
- Natclovir Ganciclovir Injection 500 MG
- Natdac Daclatasvir Tablet 60 mg
- Natdecita Decitabine Injection 50mg
- Nevimune Nevirapine Tablet 200mg
- Nublast Temozolomide Capsule 20mg
- Nublast Temozolmide Capsule 100 mg
- Nublast Temozolomide Capsule 250 mg
- Pamorelin LA Injection 11.25mg
- Pamorelin LA Injection 22.5mg
- Posid Etoposide Capsule 50 mg
- Posid Etoposide Injection 100mg
- Renoalfa Alpha Ketoanalogue Tablet
- Renolog Alpha Ketoanalogue Tablet
- Resihance Regorafenib Tablet 40 mg
- Resof Sofosbuvir & Velpatasvir 100 mg
- Resof Sofosbuvir 400mg + Velpatasvir 100mg Tablet
- Resof-L Ledipasvir (90mg) + Sofosbuvir (400mg) Tablet
- Reviro Tenofovir disoproxil fumarate Tablet 300 mg
- Ribasure Ribavirin Capsule 200 mg
- Ricovir Tenofovir disoproxil fumarate Tablet 300mg
- Ricovir EM Emtricitabine 200mg + Tenofovir disoproxil fumarate 300mg Tablet
- Ritomune Ritonavir Tablet 100mg
- Sofovir Sofosbuvir Tablet 400 mg
- Sovihep V Sofosbuvir 400mg + Velpatasvir 100mg Tablet
- Sutent Sunitinib Capsule 50mg
- Synthivan Atazanavir 300mg + Ritonavir 100mg Tablet
- Tafero Tenofovir Alafenamide Tablet 25 mg
- Tafero Em Emtricitabine,tenofovir Alafenamide Tablet
- Tafnat Tenofovir Alafenamide Tablet 25 mg
- Tamodex Tamoxifen Tablet 20 mg
- Tamodex Tamoxifen Tablet 20mg
- Tavin Tenofovir disoproxil fumarate Tablet 300 mg
- Tavin EM Emtricitabine (200mg) + Tenofovir disoproxil fumarate (300mg) Tablet
- Teevir Emtricitabine (200mg) + Tenofovir disoproxil fumarate (300mg) + Efavirenz (600mg) Tablet
- Telura Lamivudine (300mg) + Tenofovir disoproxil fumarate (300mg) + Efavirenz (600mg) Tablet
- Tenof Tenofovir disoproxil fumarate Tablet Tablet
- Tenohep Tenofovir disoproxil fumarate Tablet 300mg
- Tenolam E Efavirenz (600mg) Lamivudine (300mg) + Tenofovir disoproxil fumarate (300mg)
- Tenolam Lamivudine (300mg) + Tenofovir disoproxil fumarate (300mg) Tablet
- Tenvir Tenofovir disoproxil fumarate Tablet 300mg
- Tenvir L Lamivudine (300mg) + Tenofovir disoproxil fumarate (300mg) Tablet
- Teravir Tenofovir disoproxil fumarate Tablet 300mg
- Trasturel Trastuzumab Injection 440mg
- Trioday Lamivudine (300mg) + Tenofovir disoproxil fumarate (300mg) + Efavirenz (600mg) Tablet
- Triomune Lamivudine 150mg +Nevirapine 200mg + Stauvdine 30 mg
- Triomune Lamivudine 150 mg + Nevirapine 200mg + Stavudine 40mg Tablet
- Trustiva Emtricitabine (200mg) + Tenofovir disoproxil fumarate (300mg) + Efavirenz (600mg) Tablet
- Valgan Valganciclovir Tablet 450 mg
- Valolog Alpha Ketoanalogue Tablet
- Velasof Sofosbuvir,velpatasvir Tablet
- Velpanat Sofosbuvir (400mg) + Velpatasvir (100mg) Tablet
- Viraday Emtricitabine (200mg) + Tenofovir disoproxil fumarate (300mg) + Efavirenz (600mg) Tablet
- Virem Darunavir Tablet 600 mg
- Vivitra Trastuzumab Injection 150mg
- Vivitra Trastuzumba Injection 440 mg
- Vonavir Emtricitabine (200mg) + Tenofovir disoproxil fumarate (300mg) + Efavirenz (600mg) Tablet
- X-Vir entecavir Tablets 0.5mg
- X Vir Entecavir Tablet 1mg
- Xgeva Denosumab Injection 120mg
- Xtane Exemestane Tablet 25 mg
- Zepdon Raltegravir Tablet 400 mg
- Zobone Zoledronic Acid Injection 4mg
- Zolasta Zoledronic acid Injection 4mg
- Zoldonat Zoledronic acid Injection 4mg
- Zoldria zoledronic Injection 4 mg
- Accuzon Ceftriaxone Injection 1g
- Adrenicure Adrenaline Injection 1 mg
- Alburel - T Albumin Injection 20%
- Augpen Amoxycillin Clavulanic Acid Injection 1.2 g
- Augpen Amoxycillin & Clavulanic Acid Injection 300mg
- Augpen Amoxycillin & Clavulanic Acid Injection 600mg
- Azee Azithromycin 500 mg Tablet
- Biovorin Calcium Leucovorin Injection 15mg
- Epofit Recombinant Human Erythropoietin Alfa Injection 6000 IU
- Epofit Recombinant Human Erythropoietin Alfa Injection 10000 IU
- Erypro Recombinant Human Erythropoietin Alfa Injection 4000
- Erypro Safe Erythropoietin Alfa Injection 10000IU
- Erypro Safe Erythropoietin Alfa Injection 40000IU
- Espogen Recombinant Human Erythropoietin Alfa Injection 4000 IU
- Espogen Erythropoietin Alfa Injection 10000IU
- Erykine Erythropoietin Alfa Injection 4000IU
- Erykine Erythropoietin Alfa Injection 10000IU
- Levin Imatinib mesylate Tablet 400mg
- Ondiron Ondansetron Injection 4 mg
- Pomalid Pomalidomide Capsule 2mg
- Pomalid Pomalidomide Capsule 4mg
- Thalix Thalidomide Capsule 50 mg
- Thalix Thalidomide Capsule 100 mg
- Uromitexan Mesna Infusion 200 mg
- Uromitexan Mesna Infusion 200 mg
- Vintor Erythropoietin Alfa Injection 4000IU
- Vintor Erythropoietin Alfa Injection 6000IU
- Vintor Erythropoietin Alfa Injection 10000IU
- Wepox Epoetin Alfa Injection 2000 IU
- Wepox Erythropoietin Alfa Injection 4000IU
- Zocitab Capecitabine Tablet 500mg
- Zyrop Erythropoietin Alfa Injection 4000IU
- Zyrop Erythropoietin Alfa Injection 10000IU
- ABIRAPRO Abiraterone Acetate (500mg) Tablet
- BDRON Abiraterone Acetate 250MG Tablet
- BDRON 500MG Abiraterone Acetate Tablet
- SAMtica 500mg Abiraterone Acetate Tablet
- XBIRA-250MG Abiraterone Acetate Tablet
- Zecyte 250mg Abiraterone Acetate Tablet
- Zecyte 500mg Abiraterone Acetate Tablet
- ZELGOR Abiraterone Acetate 250mg Tablet
- Altraz Anastrozole (1mg) Tablet
- FEMISTRA 1MG ANASTROZOLE TABLET
- Anabrez Anastrozole (1mg) Tablet
- Anaday Anastrozole (1mg) Tablet
- Arimidex Anastrozole 1mg Tablet
- Armotraz Anastrozole (1mg) Tablet
- Aprecap 125/80 Aprepitant (125mg) + Aprepitant (80mg) Capsule
- Aprecap Fosaprepitant 150mg Injection
- Apretero 125/80 Aprepitant (125mg) + Aprepitant (80mg) Tablet
- Bendit Bendamustine 100mg Injection
- Bimode Bendamustine 100mg Injection
- Purplz Bendamustine 100mg Injection
- Cizumab 400 Bevacizumab (400mg) Injection
- Cizumab 100 Bevacizumab (100mg/4ml) Injection
- Bevatas 400 Bevacizumab (400mg) Injection
- Bevatas 100 Bevacizumab (100mg) Injection
- BevaciRel Bevacizumab 100mg Injection
- Bevacirel Bevacizumab 400mg Injection
- Abevmy Bevacizumab 400mg Injection
- Bryxta-400 BEVACIZUMAB 400mg Injection
- Bryxta-100 Bevacizumab 100 MG Injection
- Borviz 2.5 Bortezomib (2.5mg) Injection
- Borviz 2 Bortezomib (2mg) Injection
- Biocure-2 Bortezomib 2mg Injection
- Myezom 2 Bortezomib (2mg) Injection
- Bortenat Bortezomib 2mg Injection
- Bortenat Bortezomib 3.5mg Injection
- Bortecad Bortezomib 2mg Injection
- Cacit-500 Capecitabine 500mg Tablet
- Capegard 500 Capecitabine (500mg) Tablet
- Capiibine Capecitabine 500mg Tablet
- Capnat-500 Capecitabine 500mg Tablet
- Capsy-500 Capecitabine 500mg Tablet
- Zocitab-500 Capecitabine 500 MG Tablet
- Kemocarb Carboplatin 150mg Injection
- Kemocarb Carboplatin 450mg Injection
- Stricarb Carboplatin 450 MG Injection
- Cytoplatin-50 Cisplatin 50 Mg Injection
- Cytoplatin-10 Cisplatin 10mg Injection
- Cycloxan Cyclophosphamide 50mg Tablet
- Endoxan CYCLOPHOSPHAMIDE-50MG Tablet
- Taxotere Docetaxel 80mg Injection
- Endoxan-N 1G Cyclophosphamide (1000mg) Injection
- Endoxan N CYCLOPHOSPHAMIDE 200mg Injection
- ENDOXAN-N CYCLOPHOSPHAMIDE-500MG INJECTION
- Taxotere Docetaxel 20mg Injection
- Docetere Docetaxel 80mg Injection
- Docetere Docetaxel 20mg Injection
- Docetere Docetaxel 120mg Injection
- Docetax Docetaxel 80mg Injection
- Docetax Docetaxel 20MG Injection
- Docetax Docetaxel 120mg Injection
- DAXOTEL Docetaxel 80mg Injection
- Daxotel Docetaxel Concentrated 20mg Injection
- DAXOTEL Docetaxel 120mg/6ml Injection
- Adrim Doxorubicin Hydrochloride 10mg Injection
- Adrim Doxorubicin HCL 50mg/25ml Injection
- Adriamycin Doxorubicin Hydrochloride 50mg Injection
- Adriamycin DOXORUBICIN HYDROCHLORIDE 10 mg Injection
- Pegadria 20 Pegylated Liposomal Doxorubicin Hydrochloride Injection
- Farmorubicin Rapid Dissolution Epirubicin Hydrochloride 50mg Injection
- Farmorubicin RD Epirubicin Hydrochloride 10MG Injection
- Epithra Epirubicin Hydrochloride 50MG Injection
- Epithra Epirubicin Hydrochloride 100 Mg Injection
- Epithra Epirubicin Hydrochloride 10MG Injection
- Alrubicin Epirubicin Hydrochloride 50mg Injection
- Alrubicin Epirubicin Hydrochloride 100mg Injection
- Erlocip Erlotinib 100 Mg Tablets
- Erlocip Erlotinib 150 Mg Tablets
- Erlonat Erlotinib 100 Mg Tablets
- Erlonat Erlotinib 150 Mg Tablets
- Zyceva Erlotinib 100mg Tablet
- Zyceva Erlotinib 150mg Tablets
- Evertor Everolimus 5mg Tablets
- Evertor Everolimus 10mg Tablet
- Evermil-5 Everolimus 5 mg Tablet
- Evermil-10 Everolimus 10mg Tablets
- Rolimus-5 Everolimus 5mg Tablets
- Rolimus-10 Everolimus 10Mg Tablets
- Neukine Filgrastim 300mcg(pfs) Injection
- Neukine-300 Filgrastim 300mcg Injection
- Religrast (pfs) Filagrastim 300mcg Injection
- Nufil Safe 300mcg Injection (pfs)
- Colstim Filgrastim 300mcg (pfs) Injection
- Colstim Filgrastim 300mcg Injection
- Grastim (pfs) Filgrastim 300mcg Injection
- GRASTIM VIAL Filgrastim 300 MCG Injection
- Grafeel Filgrastim 300 Mcg Injection
- Grafeel Filgrastim 300 Mcg (pfs) Injection
- Emgrast (pfs) Filmgrastim 300 mg Injection
- Geftinat Geftinib 250mg Tablet
- Geftinat Geftinib 250mg Tablet
- Geffy Gefitinib 250mg Tablet
- Gefticip Gefitinib 250 Mg Tablet
- Gefitero Gefitinib 250mg Tablets
- Geftib Gefitinib 250 mg Tablets
- Gemita Gemcitabine 200mg Injection
- Gemita Gemcitabine 1g Injection
- Gemita GEMCITABINE 1.4gm Injection
- Cytogem Gemcitabine 1000mg Injection
- Cytogem Gemcitabine 200mg Injection
- Gemcite Gemcitabine 1gm Injection
- Gemcite Gemcitabine 200 mg Injection
- Gemtero Gemcitabine 1gm Injection
- Gemtero Gemcitabine 200mg Injection
- Gemtaz Gemcitabine 1gm Injection
- GEMTAZ Gemcitabine 200MG INJECTION
- Zoladex-3.6mg Goserelin Acetate Injection
- Zoladex LA 10.8mg Goserelin Acetate Injection
- Holoxan Ifosfamide 1 gm Injection
- Holoxan Ifosfamide 2 Gm Injection
- Imatib-400 Imatinib mesylate 400 mg Tablets
- Imatib-100 Imatinib 100 Mg Tablet
- Imanib 400mg Imatinib Tablets
- Imanib Imatinib 100 Mg Tablets
- Glivec Imatinib 400mg Tablet
- Glivec Imatinib 400mg Tablet
- Veenat-100 Imatinib 100 Mg Capsule
- Veenat-100 Imatinib 100mg Capsules
- Veenat Imatinib 400 Mg Tablets
- Imat Imatinib 100Mg Tablets
- Imat Imatinib 400mg Tablets
- Mitinab Imatinib 400 Mg Tablets
- Irinotel Irinotecan 100 Mg Injection
- Irinotel Irinotecan 40mg Injection
- Irnocam Irinotecan 40 mg Injection
- Irnocam Irinotecan 100 Mg Injection
- Lenalid Lenalidomide 5 Mg Capsules
- Lenalid Lenalidomide 10 Mg Capsules
- Lenalid 15mg Lenalidomide Capsules
- Lenalid Lenalidomide 25 Mg Capsules
- Lenangio Lenalidomide 5 mg Capsules
- Lenangio Lenalidomide 10 mg Capsules
- Lenangio Lenalidomide 25 Mg Capsule
- Letroz Letrozole 2.5mg Tablet
- Oncolet Letrozole 2.5mg Tablet
- Oreta Letrozole 2.5mg Tablets
- Fempro Letrozole 2.5 mg Tablets
- Lupride Depot Leuprolide Acetate 3.75mg Injection
- Lupride Leuprolide Acetate 11.25 Mg Injection
- Lupride Depot Leuprolide Acetate 22.5mg Injection
- Luprodex Leuprolide Acetate 3.75 Mg Injection
- Luprodex Leuprolide Acetate 11.25mg Injection
- Luprodex Leuprolide Acetate 22.5 Mg Injection
- Biotrexate Methotrexate 15mg Injection
- Biotrexate Methotrexate 2.5mg Tablet
- Biotrexate Methotrexate 500 Mg Injection
- Biotrexate Methotrexate 50mg Injection
- Megaplat Oxaliplatin 100mg Injection
- Megaplat Oxaliplatin 50 Mg Injection
- X-plat Oxaliplation 100mg Injection
- X Plat Oxaliplatin 50mg Injection
- Oxiplat Oxaliplatin 100mg Injection
- Oxiplat Oxaliplatin 50mg Injection
- Oxitan Oxaliplatin 100 Mg Injection
- Oxitan Oxaliplatino 50 Mg Injection
- Oxitan Oxaliplatino 50 Mg Injection
- Oxaltero Oxaliplatin 100 Mg Injection
- Oxaltero Oxaliplatin 50mg Injection
- Intaxel Paclitaxel 30mg Injection
- Intaxel Paclitaxel 100 Mg Injection
- Intaxel Paclitaxel 260mg Injection
- Nanoxel Paclitaxel Nanoparticle 100 Mg Injections
- Nanoxel Paclitaxel Nanoparticle 30mg Injection
- Nanoxel Paclitaxel Nanoparticle 300 Mg Injection
- Mitotax Paclitaxel 30mg Injection
- Mitotax Paclitaxel 100mg Injection
- Mitotax Paclitaxel 250mg Injections
- Mitotax Paclitaxel 300mg Injection
- Paclitax Paclitaxel IP 30mg Injection
- Paclitax Paclitaxel 100 Mg Injection
- Paclitax Paclitaxel 260 Mg Injection
- Paclitax Nab Abumin Bond Nanoparticle Paclitaxel 100Mg Injection
- Zupaxel Paclitaxel 300mg Injection
- Zupaxel Paclitaxel 260mg Injection
- Cytax Paclitaxel 30mg Injection
- Cytax Paclitaxel 100mg Injection
- Cytax Paclitaxel 260 Mg Injection
- Cytax Paclitaxel 300mg Injection
- Genexol PM Paclitaxel 100mg Injection
- TaxoNab Paclitaxel 100mg Injection
- Pegfeel (pfs) Pegylated r-Human Granulocyte Colony Stimulating Factor 6mg Injection
- Pegasta (pfs) Pegylated r-Human Granulocyte Colony Stimulating Factor 6mg Injection
- Pegstim (pfs) Pegfilgrastim 6 Mg Injection
- Pegex (pfs) Pegfilgrastim 6 Mg Injection
- PEMCURE PEMETREXED 100MG INJECTION
- Pemcure Pemetrexed 500MG Injection
- Pemnat Pemetrexed 100mg Injection
- Pemnat Pemetrexed 500mg Injection
- Reditux Rituximab 500 Mg Injection
- Reditux Rituximab (100mg) Injection
- Mabtas Rituximab 500mg Injection
- Mabtas Rituximab 100 Mg Injection
- Mabtas RA Rituximab 500 Mg Injection
- Ristova Rituximab 500 Mg Injection
- Ristova Rituximab 100mg Injection
- Maball Rituximab 500 Mg Injection
- Maball Rituximab 100 Mg Injection
- Enfiera Rituximab 100mg Injection
- Enfiera Rituximab 500mg Injection
- Rituxirel Rituximab 100mg Injection
- Rituxirel Rituximab 500 Mg Injection
- Sorafenat Sorafenib 200 Mg Tablets
- Soranib Sorafenib 200MG Tablets
- Temoside Temozolomide 250 Mg Capsule
- Temoside Temozolomide 100 Mg Capsule
- Temoside-20 Temozolomide (20mg) Capsule
- Glioz Temozolamide 250mg Capsules
- Glioz Temozolomide 100mg Capsule
- Glioz Temozolomide 20mg Capsules
- Glioz Temozolomide 100mg Capsule
- Nublast Temozolomide 100mg Capsule
- Nublast Temozolomide 250 mg Capsules
- Nublast Temozolomide 20 MG Capsules
- Hertraz Trastuzumab 440 Mg Injection
- Hertraz Trastuzumab 150 Mg Injection
- Canmab Trastuzumab 440 Mg Injection
- Canmab Transtuzumab 150mg Injection
- Herclon Trastuzumab 440mg Injection
- Vivitra Trastuzumab 150 mg Injection
- Vivitra Trastuzumab 440 Mg Injection
- Hermab Trastuzumab 440mg Injection
- Trasturel Trastuzumab 440mg Injection
- Zoldonat Zoledronic Acid 4mg Injection
- Zoldria Zoledronic Acid 4mg Injection
- Zobone Zoledronic Acid 4mg Injection
- Zobone Zoledronic Acid 4mg Injection
- Zolasta Zoledronic Acid 4mg Injection
- Exemptia Adalimumab 40mg Injection
- Arsenox Arsenic Trioxide 1mg Injection
- Azadine Azacitidine 100mg Injection
- Myaza Azacitidine 100 MG Injection
- Mamofen-10 Tamoxifen (10mg) Tablet
- Mamofen-10 Tamoxifen (10mg) Tablet
- Caditam-10 Tamoxifen 10mg Tablet
- Entax-10 Tamoxifen 10mg Tablet
- Tamoxilon-10 Tamoxifen 10mg Tablet
- Tamodex Tamoxifen 10mg Tablet
- Mamofen 20 Tamoxifen (20mg) Tablet
- Tamtero Tamoxifen 20 mg Tablet
- Caditam-20 Tamoxifen 20mg Tablet
- Caditam-20 Tamoxifen 20mg Tablet
- Tamoxilon Tamoxifen (20mg) Tablets
- Entax-20 Tamoxifen 20mg Tablet
- Tamovac-20 Tamoxifen 20mg Tablet
- Tamoxican-20 Tamoxifen 20mg Tablets
- Tamoxol -20 Tamoxifen (20mg) Tablet
- Cytotam 20 Mg Tamoxifen Citrate Tablet
- Nolvatam Tamoxifen (20mg) Tablet
- Skin Care Medicine
- 10 MG Isotretinoin Capsules IP
- 20 MG Isotretinoin Capsules IP
- 30 MG Isotretinoin Capsules IP
- 5 MG Isotretinoin Capsules IP
- 10 GM Fusidic Acid Cream IP
- 10 MG Minoxidil Tablets IP
- Tretinoin Cream USP
- 20 GM Tretinoin Cream USP
- Melacare Hydroquinone , Mometasone & Tretinoin
- Melacare Forte Hydroquinone ,Mometasone & Tretinoin Cream 20mg
- Aziderm Azelaic Acid (10% w/w) Gel
- Aziderm Azelaic Acid 20% cream
- Ezanic Azelaic Acid 10% Cream
- Rejusure 10% Azelaic Acid Suspension
- Exazel Azelaic Acid 10% Cream
- Zebor Azelaic Acid (10% w/w) Gel
- Tretiheal-0.1% Tretinoin Cream 20gm
- Tretiheal-0.05% Tretinoin Cream 20g
- Tretiheal-0.025% Tretinoin Cream 20gm
- A Ret Tretinoin Gel 0.1 %
- A-Ret Tretinoin Gel 0.05%
- A-Ret Tretinoin Gel 0.025%
- Triluma Hydroquinone + Tretinoin + Fluocinolone acetonide Cream 15 Gm
- Triluma Hydroquinone + Tretinoin + Fluocinolone acetonide Cream 15 Gm
- MELACARE Hydroquinone+Mometasone+Tretinoin Cream 25gm
- Melacare Forte Hydroquinone+Mometasone+Tretinoin 20g Cream
- Skinlite Cream 25g - Hydroquinone + Tretinoin + Momentasone Furoate Cream
- Skinlite Cream 25g - Hydroquinone + Tretinoin + Momentasone Furoate Cream
- Skinlite Cream 20gm Hydroquinoine+Tretionin+Mometasone Furoate
- Adaferin 0.1 % Adapalene gel
- Benzac AC BENZOYL PEROXIDE-2.5% Gel 20gm
- Desowen DESONIDE-0.05%W/V Lotion 30ml
- Desowen DESONIDE-0.05%W/W 10gm Cream
- Efaderm 60G Cream
- Efficort HYDROCORTISONE-0.127%W/W 10gm Cream
- Epiduo 0.1%/2.5% Gel ADAPALENE + BENZOY PEROXIDE
- Loceryl AMOROLFINE-2.5MG 30gm Cream
- PERMITE Permethrin Cream 60 gm
- Benzac AC 5 BENZOYL PEROXIDE-5%W/W Gel 30gm
- Biluma Advance Cream for Sensitive Areas 25gm
- Epiduo Forte ADAPALENE-0.3% + BENZOYL PEROXIDE-2.5% 30gm Gel
- Aklief TRIFAROTENE-0.005% 30gm Cream
- Cetaphil Cleansing & Moisturising Syndet Bar 75 gm
- Cetaphil Oily Skin Cleanser 250 ml
- Cetaphil Moisturising Cream 450 gm
- Peroclin 2.5% Benzoyl Peroxide (2.5% w/w) + Clindamycin (1% w/w) 15gm Gel
- Clenia Skin Rejuvenating Face Wash 100ml
- Aquasoft FC Advanced Facial 100gm Cream with Sun Protection
- Ilumax Ultra Advanced Skin Brightening Cream 20gm
- Elyn Eflornithine (13.9% w/w) Cream 15gm
- Peroclin 5% Benzoyl Peroxide + Clindamycin Gel 15gm
- Biosilk Moisturizing 100gm Cream
- Aqurea-HF Urea 40% Hand & Foot Cream Gel 50gm
- Ilumax Advanced Skin Lightening Cream 20 gm
- Peroduo Adapalene + Benzoyl Peroxide Gel 15gm
- Blise Face Wash 100gm Gel
- Melrio Cream Hydroquinone+Tretinoin+Fluticasone Propionate 20gm
- Opexa Dimeticone+Ascorbyl Tetraisopalmitate 10gm Gel
- Opexa Dimeticone+Ascorbyl Tetraisopalmitate 10gm Gel
- Banray Sunscreen Gel SPF 50 PA+++ 50gm
- Vaniza Polysiloxane & Silicone Dioxide Gel 10gm
- Pacroma XL Pimecrolimus (1% w/w) 30gm Cream
- Cernos Testosterone Gel 5g
- Biosilk Max Intense Moisturizing Cream 150gm
- AVEXA 100gm Cream
- Ivrea 1 Ivermectin 1% w/w Cream
- Civaderm XL 1% Ciclopirox (1% w/w) Cream
- Avexa Lotion 150ml
- New Aquasoft CV Daily Anti-Itch Lotion 100ml
- Sunstop Acne Spf 80 Pa++++ Gel 50gm
- Vaniza Gold Gel 15gm
- Candid-B Cream Beclometasone + Clotrimazole 20gm
- Candid-B Lotion Beclometasone + Clotrimazole 30ml
- Konzert Sertaconazole 2% Cream 20g
- Candid CLOTRIMAZOLE Cream 20gm
- Deriva MS Aqueous Adapalene Gel 15gm
- Deriva-CMS Adapalene + Clindamycin Gel 20gm
- Deriva-BPO Adapalene + Benzoyl Peroxide Gel 20gm
- Candid Dusting Powder Clotrimazole 250gm
- Momate Mometasone 20gm Cream
- Momate-F Mometasone + Fusidic Acid Cream 15gm
- Momate-XL Mometasone Cream 40gm
- Momate T Tazarotene + Mometasone Cream 15gm
- Supirocin Ointment Mupirocin (2% w/w) 5gm
- Supirocin F Fluticasone Propionate + Mupirocin Ointment 10gm
- Supirocin Mupirocin Cream 10gm
- Supirocin-B Plus Ointment Mupirocin + Beclometasone 5gm
- Supirocin Lipogel Mupirocin 15gm
- Imiquad Cream Imiquimod (12.5mg) (0.25gm Each)
- Scaboma Lotion Lindane 100ml
- Tacroz Ointment Tacrolimus (0.03% w/w) 20gm
- Tacroz Forte Tacrolimus 20gm Ointment
- Tacroz Forte XL Tacrolimus Ointment 40gm
- Tacroz Forte Tacrolimus Solution 20ml
- Elovera Pro Moisturising Cream 75gm
- Aprezo 20 Apremilast (20mg) Tablet
- Aprezo 10 Apremilast (10mg) Tablet
- Aprezo-30 Apremilast 30mg Tablet
- Excela Moisturiser for Oily & Acne Prone Skin 50gm
- Saslic DS Foaming Face Wash 60ml
- Saslic Salicylic Acid Foaming Face Wash 60ml
- Saslic Salicylic Acid Foaming Face Wash 60ml
- VCX Vitamin C, E & Ferulic Acid Serum 10ml
- Rivela Sunscreen SPF 50 Lotion 50ml
- VC 15 Vitamin C Serum 15ml
- Cetafresh Cleansing Lotion 200ml
- Rivela Tint Sunscreen SPF 50 Lotion 50ml
- Excela Rich Facial Hydrating Lotion 50gm
- VC GroF Cellglow Liquid 30gm
- Lumacip HYDROQUINONE Cream 30 gm
- Lumacip Hq Depigmenting Complex 30gm
- Acnedap Plus Dapsone + Adapalene Gel 15gm
- Excela Max Moisturiser for Dry & Itchy Skin 200gm
- Depiwhite Skin Brightening Cream 15ml
- Antoxid-HC Antioxidant Capsule
- Ezinapi Plus Cream 50gm
- Antoxid Antioxidant Capsule
- Venusia Moisturizing Cream 100gm
- Atarax Cream For Distressed Dry And Itchy Skin 100gm
- Vitix Skincare Depigmentation Gel 50ml
- Venusia Acne Control Moisturizer 75gm
- Atarax Anti-Itch Lotion 100ml
- Revibra C10 Pure & Bioactive Vitamin C Cream
- Revibra A15 Pure Retinol Vitamin A Cream
- Venusia Max Intensive Moisturizing Lotion 500gm
- Venusia DERM Moisturizing Cream 75gm
- Venusia DERM Moisturizing Cream 75gm
- Venusia DERM Moisturizing Cream 75gm
- Venusia DERM Moisturizing Cream 75gm
- Venusia DERM Moisturizing Cream 75gm
- Venusia Ceraplus Cream 100gm
- Revilus Kz Ketoconazole + Zinc pyrithione Lotion 100ml
- Venusia Sun Tint Velvet Touch Sunscreen Gel SPF 115+ PA++++
- Ebernet Eberconazole (1% w/w) Cream 60gm
- Ebernet Plus Eberconazole (1% w/w) Cream
- Venusia Ureka Cream 100gm
- Ebernet-M Eberconazole + Mometasone Cream
- Ebernet Lot Eberconazole 1% Cream
- Fixtral SB Itraconazole 100mg Capsule
- Venusia Moisturising Lotion Hydrates Dry & Rough Skin 200ml
- Fair Eye Cream Advanced Dark Circle Care 15gm
- MemBio Hydra Bio-Membrane Skin Repair Cream
- Melgain Deca Peptide (1mg/ml) Lotion 10ml
- Fair Insta Skin Lightening Cream 20gm
- Livalite Lotion 30ml
- Nioret Nano Tretinoin (0.025% w/w) Gel
- Zyclin Nano Clindamycin Gel 15gm
- Litearm Lotion 30ml
- Livafin CP Amorolfine Cream 50g
- Danclear Ketoconazole + Zinc Cream 50g
- MMS Mometasone Cream 30gm
- Naftifast Naftifine Hydrochloride Cream 30gm
- ZyKT Ketoconazole Cream 15gm
- Zyluli Luliconazole 1% Cream 30gm
- Fair Insta SPF 30 Cream 20gm
- Solecross Sun Block Sunscreen Lotion SPF 50 PA++++
- Adalene Nanogel Adapalene+ Clindamycin Gel 15gm
- Clop S Nano Clobetasol + Salicylic Acid Lotion 20ml
- Itchcam Lotion 60ml
- Aloekin Lotion 100ml
- Aziderm Azelaic Acid (10% w/w) Gel
- Glyco 12 Glycolic Acid 12% 30gm Cream
- Glyco 6 Glycolic Acid 6% 30g Cream
- Aziderm 10% Azelaic Acid Cream 15gm
- Kojivit Ultra Gel Derma Care 30gm
- Aziderm 20% Azelaic Acid Gel 15gm
- Kojivit Plus Skin Lightening & Brightening Gel 15gm
- Aziderm Plus Azelaic acid+glycolic acid Cream 15g
- Kojivit Gel with Kojic Acid, Arbutin, Mulberry Extract & Vitamin E
- Nilac Clindamycin + Nicotinamide Gel 20g
- Nilac A Adapalene + Clindamycin Gel 20gm
- Calosoft-AF Calamine Lotion 100ml
- Secalia Glycerin Cream for Dry Skin 100gm
- Calosoft Plus Moisturising Lotion 100ml
- Secalia Glycerin Lotion 100ml
- Monoguard Sertaconazole Cream 50gm
- OneTone Plus Cream 20gm
- Phytoral Ketoconazole Ointment 30gm
- Phytoral Ketoconazole Ointment 30gm
- Phytoral B Beclometasone + Ketoconazole Cream 15gm
- Isotane 20 Isotretinoin (20mg) Capsule
- Photoban 30 Aquagel 60gm
- Lycor 1% Hydrocortisone Cream 15gm
- Ga - 6 Glycolic Acid Cream 30g
- Blemguard Face Serum 30 ml
- CosmoQ OC Moisturizing Gel 60g
- Nevlon Moisturizing Cream 100gm
- Nevlon Moisturizing Cream 100gm
- Klmc 20 20% Vitamin C Serum 15ml
- Zydip-C Beclometasone + Clotrimazole Lotion 50ml
- Sebozit Salicylic Gel 30gm
- Glutaderm Skin Tablet
- Melboost Deca Peptide 5mg Solution 5ml
- CosmoQ Moisturizing Cream 50gm
- Ga -12 Glycolic Acid Cream 30gm
- Nevlon Anti-Itch Cream 100gm
- Tyrolite Cream 15gm
- Melapik -HQ Hydroquinone Cream 20gm
- Nioclean AD Adapalene + Clindamycin Gel 15gm
- Glutaderm Plus Tablets
- Zuvimax 0.2% Pure Retinol Cream 20gm
- Nevsoft Cleansing Cetyl Alcohol + Stearyl Alcohol Lotion 125ml
- Nevlon-Max Cream 150gm
- Kojitin Ultra Emulgel 20gm
- Melapik Plus Hydroquinone + Tretinoin + Fluocinolone Cream 20gm
- Trunex MS Aqueous Tretinoin Gel 15gm
- Kojitin Skin Lightening Emulgel 15gm
- Strianil Anti-Stretch Mark Cream 50gm
- Lulizol Luliconazole Cream 30g
- Eberfine Eberconazole Cream 30gm
- Cutiheal Hydrocortisone 1% Cream 15g
- Cutiheal Hydrocortisone 1% Cream 15g
- Nevlon Caloe Lotion 100ml
- Xerolene Cream For Thick, Rough, Dry & Cracked Heels 50gm
- Tecum Tacrolimus 0.1% Ointment 10gm
- Sofidew Resyl Lotion 50ml
- Canrolfin Amorolfine (0.25% w/w) Cream 30gm
- Lulizol XL Luliconazole 1% Cream 50g
- Zycozol Ciclopirox 1% Cream 30g
- Ofacitix Tofacitinib 2% Ointment 15g
- Mfsone Mometasone Cream 30g
- Niogenta F Beclometasone + Fusidic Acid Cream 20g
- Episert Sertaconazole Cream 30g
- Niosalic Clobetasol + Salicylic Ointment 20gm
- KLMC-20 GEL 20gm
- Aprezo Apremilast 30 Mg Tablet
- Moisawave Moisturizing Cream 100g
- ScarEnd Gel 15gm
- Moisawave Moisturising Lotion 250ml
- ScarEnd Silicone Gel 15gm
- Alograce Moisturising Cream 50gm
- Calapure-A Lotion 100ml
- Momstar Mometasone Cream 15g
- Itragreat 100 Itraconazole Capsule
- Hicope Lotion
- Lulibrut Luliconazole Cream 50gm
- Zitblow-10 Isotretinoin 10mg Soft Gelatin Capsule
- Ketostar Ketoconazole Cream 30gm
- Skinbless Cream Enriched with AA2G (Vitamin C) 20gm
- Acnestar Clindamycin & Nicotinamide Gel 22gm
- Pain Killer And Muscle Medicines
- 300 MG Gabapentin Capsules IP
- 300 MG Pregabalin Capsule
- 100 MG pharma Tablets IP
- 130 MG pharma Tablets USP
- Pharma Tablets USP
- 100 MG pharma Tablets
- 50 MG Vildagliptin Tablets IP
- Bonmax PTH Injection 750mcg
- Botox Botulinum Toxin Injection 100 mg
- Botox Clostridium botulinum type A toxin-haemagglutinin complex (200IU) Injection
- Caprin Heparin Injection 25000IU
- Gemtide Teriparatide Injection 600 mcg
- Lox Lidocaine Injection 2%
- Lox Lidocaine & Adrenaline Injection 2%
- Lox Lignocaine Topical Injection 4%
- Lox Lidocaine Spray 10%
- Mezolam Midazolam Injection 10 mg
- Neorof Propofol Injection 1%
- Terifrac Teriparatide Injection 750mcg
- Anti Infection Medicines
- 75 MG Oseltamivir Capsules IP
- Ivermenctin Tablets
- Ivermectin Tablets USP
- Bimat Bimatoprost Eye Drop
- Bionesp Darbepoetin alfa Injection 25mcg
- Botox Botulinum Toxin Injection 50 mg
- C Tri Ceftriaxone Injection 1g
- Casfung Caspofungin Injection 50 mg
- Casfung Caspofungin Injection 70mg
- Cefpraz Cefoperazone & Sulbactam Injection 500 mg
- Ciprox Ciprofloxacin Tablet 500mg
- Clariwok Clarithromycin Injection 500mg
- Dalacin C Clindamycin & Disodium edetate Injection 600mg
- Emtig Tigecycline Injection 50mg
- Globucel Immunoglobulin Infusion 5gm
- Kanamac Kanamycin Injection 750mg
- Kanamac Kanamycin Injection 1000mg
- Magnex Forte Cefoperazone & Sulbactam Injection 1.5 g
- Mega CV Amoxycillin & Clavulanic Acid Injection 1.2gm
- Monocef Ceftriaxone Injection 1000mg
- Montaz Ceftriaxone & Tazobactum Injection 1g
- Oflox OfIoxacin Infusion 2mg
- Phosome Liposomal Amphotericin B Injection 50mg
- Picasa Posaconazole Oral Suspension 40mg
- Powercef Ceftriaxone Injection 1g
- Rhoclone Anti Rh D Immunoglobulin Injection 150mcg
- Rhoclone Anti Rh D Immunoglobulin Injection 300mcg
- Vingraf Tacrolimus Capsule 0.5 mg
- Vingraf Tacrolimus Capsule 1mg
- Vingraf Tacrolimus Capsule 2mg
- Voraze Voriconazole Tablet 200mg
- Vorier Voriconazole Tablet 200mg
- Voritek Voriconazole Tablet 200 mg
- Xone Ceftriaxone Injection 1g
- Xylistin Colistimethate Sodium Injection 1M
- Xylistin Colistimethate Sodium Injection 4.5M
- Rozet Azithromycin 500mg Tablet
- Rozet LB 250mg Azithromycin (250mg) + Lactic acid bacillus Tablet
- Allegra-120 Fexofenadine (120mg) Tablet
- Allegra 180 Fexofenadine (180mg) Tablet
- Allegra-M Montelukast (10mg) + Fexofenadine (120mg) Tablet
- Lupins Benz Pearls Benzonatate (100mg) Capsule
- Montair-LC Levocetirizine (5mg) + Montelukast (10mg) Tablet
- Anti-Biotics Medicines
- 18 MG pharma Hydrochloride Tablets
- 5 MG Prednisolone Tablets IP
- 200 MG Hydroxychloroquine Sulphate Tablets IP
- 500 MG Fenbendazole Tablet
- EMSYL Etamsylate Injection 2ML
- Enoxatroy Enoxaparin Injection 20mg
- Enoxatroy Enoxaparin Injection 40mg
- Enoxatroy Enoxaparin Injection 60mg
- Klacid IV Clarithromycin Injection 500MG
- Linetic Linezolid 600mg Infusion
- Loxazin Levofloxacin Tablet 500mg
- M Penem Meropenem Injection 1000 mg
- Magnova Cefepime & Tazobactum Injection 1g
- Mega-CV Amoxycillin Clavulanic Acid Injection 600mg
- Mero Meropenem Injection 1gm
- Merobax Meropenem Injection 1gm
- Merocrit Meropenem Injection 1000mg
- Meronem Meropenem Injection 1000mg
- Merotec Meropenem Injection 1000mg
- Moxicip Moxifloxacin Infusion 400 mg
- Piptaz Piperacillin & Tazobactum Injection 2.25 g
- Piptaz Piperacillin & Tazobactum Injection 4.5 g
- Pipzo Piperacillin & Tazobactum Injection 2.25gm
- Pipzo Piperacillin & Tazobactum Injection 4.5gm
- Poly Polymyxin B Injection 500000IU
- Regenta Gentamicin Injection 80mg
- Targocid Teicoplanin Injection 200mg
- Targocid Teicoplanin Injection 400mg
- Taxim Cefotaxime Injection 1g
- Taxim Cefotaxime Injection 250mg
- Taxim Cefotaxime Injection 500mg
- Tazact Piperacillin & Tazobactum Injection 2.25gm
- Tazact Piperacillin & Tazobactum Injection 4.5gm
- Tazar Piperacillin & Tazobactum Injection 2.25 gm
- Tazar Piperacillin & Tazobactum 4.5 gm
- Tazilin Piperacillin & Tazobactum 4.5 gm
- Tazomac Piperacillin & Tazobactam Injection 2.25 gm
- Tazomac Piperacillin & Tazobactum Injection 4.5gm
- Tazact Piperacillin & Tazobactum Injection 2.25gm
- Tazotum Piperacillin & Tazobactam Injection 4.5gm
- Ticocin Teicoplanin Injection 400 mg
- Tigebax Tigecycline Injection 50 mg
- Troyperazone Cefoperazone & Sulbactam Injection 1.5 gm
- Vanking Vancomycin Infusion 1gm
- Viatran Cefoperazone & Sulbactam Injection 1.5 gm
- Xylistin Colistimethate Sodium Injection 2M
- Zienam Imipenem & Cilastatin Injection 500mg
- Zostum Cefoperazone & Sulbactam Injection 1.5g
- Zostum Cefoperazone & Sulbactam Injection 1g
- Anti-Asthma And Anti-Copd Medicines
- 200 MG Tanpentadol Extended Release Tablets
- Salbutamol Inhalation IP
- 400 MG Doxofylline Tablets IP
- 500 MG Salmeterol And Fluticasone Propionate Powder For Inhalation IP
- Budesonide-Formoterol Inhalation Powder
- Budecort Budesonide Respules 0.5mg
- Duolin Levosalbutamol & Ipratropium Respules
- Neo-Drol Methylprednisolone Injection 500 mg
- Pyrolate Glycopyrrolat Injectione 0.2mg
- Symbicort 320mcg/9mcg Formoterol + Budesonide Turbuhaler
- Symbicort 160mcg/4.5mcg Turbuhaler
- Symbicort 80mcg/4.5mcg Formoterol + Budesonide Turbuhaler
- Formonide Forte Formoterol + Budesonide Respicap
- Formonide Forte 12mcg/400mcg Formoterol + Budesonide Inhaler
- Formonide 200 Formoterol (6mcg) + Budesonide (200mcg) Inhaler
- Formonide 400 Formoterol (6mcg) + Budesonide (400mcg) Respicaps
- Formonide 200 Formoterol (6mcg) + Budesonide (200mcg) Capsule
- Foracort Formoterol+Budesonide Inhaler 200 Mg
- Foracort 0.5mg Formoterol + Budesonide Respules (2ml Each)
- Foracort 200 Formoterol + Budesonide Rotacap
- Foracort 400 Formoterol + Budesonide Rotacaps
- Foracort 400 Formoterol + Budesonide Inhaler
- Foracort 200 Synchrobreathe Inhaler
- Foracort 400 Synchrobreathe Inhaler
- Foracort G 400 Glycopyrrolate + Formoterol + Budesonide Rotacap
- Foracort 100 Formoterol + Budesonide Inhaler
- Foracort Junior Formoterol + Budesonide Inhaler
- Foracort G 25mcg/12mcg/400mcg Capsule
- Foracort 200 Formoterol + Budesonide Autohaler
- Asthalin Salbutamol 100mcg Inhaler
- Asthalin Salbutamol (200mcg) Rotacap Capsule
- Asthalin Salbutamol (2mg/100ml) Syrup
- Asthalin Salbutamol (2.5mg) Respules (2.5ml Each)
- Aerocort Levosalbutamol + Beclometasone Inhaler
- Aerocort Levosalbutamol + Beclometasone Rotacap
- Aerocort Forte Levosalbutamol + Beclometasone Rotacap
- Duolin Levosalbutamol + Ipratropium Inhaler
- Combimist-L Levosalbutamol + Ipratropium Respules 2.5ml
- Combimist-L Levosalbutamol + Ipratropium Respicap
- Combimist-L CFC Free Inhaler
- Combimist LD (2.5ml Each) Respules
- Combinase AQ Fluticasone Propionate + Azelastine Nasal Spray
- Budecort 1mg Respules (2ml Each)
- Budecort 200 Budesonide (200mcg) Inhaler
- Budecort 100 Budesonide (100mcg) Inhaler
- Budecort 200 Budesonide (200mcg) Rotacap
- Budecort 400 Budesonide (400mcg) Rotacaps
- Budenase AQ Budesonide (100mcg) Nasal Spray
- Fluticone Fluticasone Propionate (0.05% w/v) Nasal Spray
- Tiova Inhaler Tiotropium Bromide 9mcg Inhaler
- Tiova Tiotropium (18mcg) Rotacap
- Trelegy Ellipta Powder Fluticasone Furoate+ Umeclidinium+ Vilanterol For Inhalation
- Asthalin 100mcg Salbutamol Albuterol Inhaler
- Asthalin Salbutamol (200mcg) Rotacap Capsule
- Levolin Salbutamol 50mcg Autohaler
- Asthavent Salbutamol 100mcg Inhaler
- Azmasol Salbutamol 100mcg/puff Inhaler
- Ventorlin Salbutamol (100mcg) Inhaler CFC Free
- Derihaler Salbutamol 100mcg Inhaler
- Salbair-B Salbutamol 100mcg+Beclometasone 50mcg Inhaler
- BUDETROL 200 Formoterol + Budesonide Inhaler
- Maxiflo 250 Formoterol + Fluticasone Propionate Inhaler
- Forglyn Formoterol + Glycopyrrolate CFC Free Inhaler
- Digihaler FB 200 Formoterol + Budesonide Inhaler
- Arthritis Medicines
- 80 MG Febuxostat Tablets
- Collagen Peptide Glucosamine Sulphate Chondrotin Sulfate Ginger Root Extract And Vitamin C Tablets
- Kuka Cough Tablets
- 100 MG Minocycline Hydrochloride Tablets USP
- Dynapar AQ Diclofenac Injection 75mg
- Infimab Infliximab Injection 100 mg
- Intacept Etanercept Injection 25 mg
- Intacept Etanercept Injection 50 mg
- Remicade Infliximab Injection 100mg
- Weight Loss Medicines
- Cardiac And Diabetic Medicines
- 10 MG Amlodipine Tablets IP
- Atorvastatin Tablets IP
- Clonidine Hydrochloride Tablets IP
- 10 MG Atorvastatin Tablets IP
- 1000 MG Metformin Hydrochloride Prolonged Release Tablets IP
- 10 MG Rosuvastatin Tablets IP
- 50 MG Sitagliptin Tablets IP
- 80 MG Telmisartan Tablets IP
- 90 MG Ticagrelor Tablets IP
- 16 MG Cardesartan Cilexetil Tablets
- Do-min Dopa-mine Injection 200mg
- Dotamin Dobutamine Injection 250mg
- Enclex Enoxaparin Injection 40 mg
- Evaparin Enoxaparin Injection 300mg
- Flexbumin Infusion Albumin 20%
- Frusemide Furosemide Injection 2ml
- Icikinase Streptokinase Injection 1.5MIU
- Lmwx Enoxaparin Injection 20mg
- Lmwx Enoxaparin Injection 40 mg
- Lmwx Enoxaparin Injection 60 mg
- Lonopin Enoxaparin Injection 20 mg
- Lonopin Enoxaparin Injection 40mg
- Lonopin Enoxaparin Injection 60mg
- Lupenox Enoxaparin Injection 20mg
- Lupenox Enoxaparin Injection 40mg
- Lupenox Enoxaparin Injection 60mg
- Neticol Citicoline Injection 250mg
- Termin Mephentermine Injection 30mg
- U Frag Urokinase Injection 5000IU
- Mounjaro KwikPen Tirzepatide (7.5mg) Injection
- Mounjaro KwikPen Tirzepatide (15mg) Injection
- Mounjaro KwikPen Tirzepatide (12.5mg) Injection
- Mounjaro KwikPen Tirzepatide (10mg) Injection
- Xarelto Rivoraxaban 20 Mg Tablet
- Xarelto 15mg Rivaroxaban Tablets
- Xarelto Rivaroxaban 10mg Tablets
- Telma H Telmisartan (40mg) + Hydrochlorothiazide (12.5mg) Tablet
- Telma 20 Telmisartan (20mg) Tablet
- Telma 80 Telmisartan (80mg) Tablet
- Telma-CT 40/6.25 Telmisartan (40mg) + Chlorthalidone (6.25mg) Tablet
- Telma-CT 40/12.5 Telmisartan (40mg) + Chlorthalidone (12.5mg) Tablet
- Telma-Beta 25 Telmisartan (40mg) + Metoprolol Succinate (25mg) Extended Release Tablets
- Telma 80-AM Telmisartan (80mg) + Amlodipine (5mg) Tablet
- Telma-CT 80/12.5 Telmisartan (80mg) + Chlorthalidone (12.5mg) Tablet
- Telmavas 40 Telmisartan (40mg) Tablet
- Telma-NB Nebivolol (5mg) + Telmisartan (40mg) Tablet
- Telma-CT 80/6.25 Telmisartan (80mg) + Chlorthalidone (6.25mg) Tablet
- Telma ACT 40mg/5mg/6.25mg Tablet
- Telmaxx 50 Telmisartan (40mg) + Metoprolol Succinate (50mg) Tablet ER
- Rozat 10 Rosuvastatin (10mg) Tablet
- Rozat 5 Rosuvastatin (5mg) Tablet
- Rozat-F Fenofibrate (160mg) + Rosuvastatin (10mg) Tablet
- Rozat 20 Rosuvastatin (20mg) Tablet
- Rozat CV Rosuvastatin (10mg) + Clopidogrel (75mg) Capsule
- Rozat-F 5 Fenofibrate (160mg) + Rosuvastatin (5mg) Tablet
- Rozat 40 Rosuvastatin (40mg) Tablet
- Endobloc 5 Ambrisentan (5mg) Tablet
- Pulmonext 5mg Ambrisentan (5mg) Tablets
- Ambrican 5 Ambrisentan (5mg) Tablet
- Endobloc 10 Ambrisentan (10mg) Tablet
- Pulmonext 10 Ambrisentan (10mg) Tablet
- Ecosprin 75 Aspirin (75mg) Tablet
- Ecosprin 150 Aspirin (150mg) Tablet
- Ecosprin AV 75/20 Atorvastatin + Aspirin Capsule
- Ecosprin Gold 20 Aspirin + Atorvastatin + Clopidogrel Capsule
- Ecosprin Gold 20 Aspirin + Atorvastatin + Clopidogrel Capsule
- Ecosprin Gold 10 Aspirin + Atorvastatin + Clopidogrel Capsule
- Ecosprin AV 75/40 Atorvastatin + Aspirin Capsule
- Ecosprin AV 150/20 Atorvastatin + Aspirin Capsule
- Ecosprin Gold 40 Aspirin + Atorvastatin + Clopidogrel Capsule
- Atorlip F Atorvastatin + Fenofibrate Tablet
- Atorlip Atorvastatin 80 Mg Tablets
- Atorlip Atorvastatin 20MG Tablets
- Atorlip Gold 20 Aspirin + Atorvastatin + Clopidogrel Capsule
- Atorlip 5 Atorvastatin (5mg) Tablet
- Atorlip 40 Atorvastatin (40mg) Tablet
- Atorlip ASP 10 Atorvastatin + Aspirin Capsule
- Nephrology Drugs
- 1 MG Sirolimus Tablets
- Sirolimus Tablets
- Cell CEPT Mycophenolate mofetil Tablet 500mg
- Cresp Darbepoetin alfa Injection 25 mcg
- Cresp Darbepoetin alfa Injection 40mcg
- Cresp Darbepoetin alfa Injection 60mcg
- Cresp Darbepoetin alfa Injection 10mcg
- Cresp Darbepoetin alfa Injection 200mcg
- Cresp Darbepoetin alfa Injection 500 mcg
- Darbecure Darbepoetin alfa Injection 25mcg
- Darbecure Darbepoetin alfa Injection 40mcg
- Darbotin Darbepoetin alfa Injection 25mcg
- Darbotin Darbepoetin alfa Injection 40mcg
- Darbotin Darbepoetin alfa Injection 60mcg
- Dargen Darbepoetin alfa Injection 25 mcg
- Dargen Darbepoetin alfa Injection 40mcg
- Dargen Darbepoetin alfa Injection 60mcg
- Mircera Methoxy polyethylene glycol-epoetin beta Injectiom 100mcg
- Neo Recormon Erythropoietin Beta Injection 5000 IU
- Relipoietin Erythropoietin Alfa Injection 2000IU
- Relipoietin Erythropoietin Alfa Injection 4000IU
- Relipoietin Erythropoietin Alfa Injection10000IU
- Renocel Erythropoietin Alfa Injection 4000IU
- Renocel Erythropoietin Alfa Injection 10000IU
- Rpo Erythropoietin Alfa Injection 4000IU
- Rpo Erythropoietin Alfa Injection 6000IU
- RPO Erythropoietin Alfa Injection 10000 IU
- Sevcar Sevelamer Tablet 400 mg
- Sevcar Sevelamer Tablet 800 mg
- Vintor Erythropoietin Alfa Injection 2000IU
- Stomach Generic Medicines
- Pantoprazole Gastro-Resistant And Domperidone Prologned-Release Capsules IP
- 750 MG Levetiracetam Tablets IP
- 135 MG Mebeverine Hydrochloride Tablets IP
- Rabeprazole Sodium (EC) And Levosulpiride (SR) Capsules
- 550 MG Rifaximin Tablets
- Eema HMG Menotrophin Injection 150IU
- Eema HP Injection 5000 IU
- Drotakop Drotaverine Injection 40mg
- Eema Fsh Urofollitropin Injection 75IU
- Eema HP Injection 10000 IU
- Enema Bp Sodium Acid Phosphate 100 Ml
- Hepamerz L-Ornithine L-Aspartate Infusion 5 mg
- Igwok Immunoglobulin 5gm Infusion
- Immunorel Human Gamma Globulin Infusion5gm
- Immunorel Human Gamma Globulin Injection 10mg
- Metharmed Methylergometrine Injection
- Mofetyl Mycophenolate mofetil Tablet 500 mg
- Mofetyl -S Mycophenolate sodium Tablet 360mg
- Mofilet Mycophenolate mofetil Tablet 250 mg
- Mofilet Mycophenolate mofetil Tablet 500 mg
- Mofilet S Mycophenolate sodium Tablet 360mg
- Mycept Mycophenolate mofetil Capsule 250 mg
- Mycept Mycophenolate mofetil Tablet 500 mg
- Mycept -S Mycophenolate sodium Tablet 360 mg
- Mycofit Mycophenolate mofetil Tablet 250 mg
- Mycofit Mycophenolate mofetil Tablet 500mg
- Nodosis Sodium Bicarbonate Tablet 500 mg
- Nodosis - DS Sodium Bicarbonate Tablet 1000mg
- Pangraf Tacrolimus Capsule 0.5 mg
- Pangraf Tacrolimus Capsule 1mg
- Pangraf Tacrolimus Capsule 2mg
- Prograf Tacrolimus Capsule 0.5mg
- Prograf Tacrolimus Hard Capsule 1mg
- Proluton Depot Hydroxy progesterone Injection 500 mg
- Rapacan Sirolimus Tablet 1mg
- Rebez IV Rabeprazole Injection 20mg
- Renodapt Mycophenolate mofetil Tablet 500mg
- Renodapt S Mycophenolate sodium Tablet 360mg
- Siromus Sirolimus Tablet 1mg
- Tacrograf Tacrolimus Capsule 0.5 mg
- Tacrograf Tacrolimus Capsule 0.25 mg
- Tacrograf Tacrolimus Capsule 1mg
- Tacrograf Tacrolimus Capsule 2mg
- Takfa Tacrolimus Capsule 0.5 Mg
- Takfa Tacrolimus Capsule 1mg
- Takfa Tacrolimus Capsule 2mg
- Eye Drops
- Anti-Smoking And Alcholic Medicine
- Adhd Drugs
- Anti Malerial Drugs
- Hair Care Medicines
- 0.5 MG pharma Tablets
- 2.5 MG Minoxidil Tablets IP
- Ionax T Scalp Solution 60ml
- Morr F 5% Minoxidil+Finasteride Solution
- Morr F 10% Minoxidil+Finasteride Solution
- Morr 5% Minoxidil Solution
- Morr 10% Minoxidil Solution
- Morr 12.5% Minoxidil Solution
- Mintop Forte 5% Minoxidil Solution
- Mintop 10% MINOXIDIL Solution
- Mintop 5% Minoxidil Solution
- Trilast Stem Intensive Hair Serum 60ml
- Tricosilk Pro Hair Solution Minoxidil + Finasteride (60ml)
- Trilast Hair Solution 60ml
- Tugain Men 5% Minoxidil + Finasteride Solution 60ml
- 8X-KT Shampoo Ketoconazole + Zinc pyrithione 60ml
- Tugain Eva Solution Minoxidil 60ml
- Tugain F+ 10 Solution Minoxidil + Finasteride 60ml
- Tugain 2% Minoxidil Solution 60ml
- Adgain Hair Enzyme Booster 60ml
- Adgain Grof Cellglow Liquid 30gm
- Ketocip 2% Ketoconazole Shampoo 100ml
- Bontress PRO+ Scalp Serum 60ml
- Mintop Pro+ Hair Regrowth Serum 50ml
- Mintop Gain 5 + Hair Regrowth Formula Kit
- Finax Finasteride 1mg Tablets
- Mintop Minoxidil (2.5mg) Tablet
- Mintop Eva Solution Minoxidil 5% 60ml
- Revilus Soflets with Multivitamins, Micronutrients & Minerals
- Mintop Yuva MINOXIDIL 5% Solution
- Mpower F Minoxidil + Finasteride Solution 60ml
- Mpower 5% Minoxidil Solution 60ml
- Mpower 2% Minoxidil Solution 60ml
- BTN Ultra Tablet For Hair Growth
- Zycafy Liquid Scalp Solution 100ml
- BTN Forte Biotin (10mg) Tablets
- Coverit 5% Minoxidil Solution 60ml
- Coverit 10% Minoxidil Solution 60ml
- Coverit Plus Spray Minoxidil + Finasteride 100ml
- Coverit 2% Minoxidil Topical Solution 60ml
- Phytoral SP Scalp Lotion 60ml
- Imxia 5 Minoxidil Solution 60ml
- Imxia F 5% Minoxidil + Finasteride Solution 60ml
- Imxia 10% Minoxidil Solution 60ml
- Kenz-Sal Ketoconazole + Salicylic Acid Lotion 60ml
- Extend Hair Tablet
- Imxia XL Serum 60ml
- Kenz Plus Ketoconazole + Zinc Lotion 100ml
- Extend Forte Tablet for Hair supplement
- Xaliderm Scalp Solution 50ml
- Chekfall-F 5% Minoxidil + Finasteride Solution 60ml
- Chekfall 5% Minoxidil Topical Solution 60ml
- Chekfall-F 10% Minoxidil + Finasteride Solution 60ml
- Chekfall 10% Minoxidil Solution 60ml
- Chekfall Minoxidil 2.5mg Tablet
- Ketostar Anti Dandruff Ketoconazole + Zinc Lotion 50ml
- Hairbless Tablet (Minerals,Amino acid with Biotin and Niacin
- Hairbless Diva Nutraceutical Tablet for Women
- Anti Anxiety Medicines
- Veterinary Medicines
- 150 MG Fenbendazole Tablets
- Sure Rab Rabies vaccine Vaccine 2.5IU
- NP E15 Nimesulide, Paracetamol & Serratiopeptidase Vet Bolus
- Rakshamin -F Bolus
- Super Gesic flunixin meglumine Injection
- C4ALL Cefquinome Sulphate Injection 500mg
- Enrox Enrofloxacin Injection 10%
- Estrofit Combination of HMTBa Minerals , Vitamins Bolus
- Melambic-p Maloxicam & Paracetamol Tablet
- Moral Marbofloxacin Injection IP
- Moxel Amoxycillin & Cloxacillin Injections 3000 mg
- Moxel Amoxycillin & Cloxacillin Injection 4000mg
- Moxel Forte Amoxycillin Sodium & Sulbactam Sodium Injection 4.5 gm
- Rumen FS Bolus 4's
- Alvifen Plus Bolus
- Alviphos Butophosphan & Cyanocobalamin Injection 100 ml
- Alviphos Butaphosphan And Cyanocobalamin Injection 30 ml
- Banminth Forte Morantel Citrate Bolus
- Banminth Morantel citrate Tablet 10s
- Bioboost Probiotics , Amino Acid & Liver Extract Bolus
- Calject Calcium Borogluconate Injection 450ml
- Dectomax Doramectin Injection 20ml
- Dectomax Doramectin Injection 50ml
- Lutalyse Dinoprost Tromethamine Injection 10 ml
- Lykacetin Chloramphenicol Sodium Succinate Injection 3gm
- Lymec Ivermectin Injection 100 ml
- Nil -O- Pain Plus Meloxicam & Paracetamol Injection 100 ml
- Oxytetracycline LA Injection
- Oxytetracycline LA Dihydrate Injection 50ml
- Resfur Ceftiofur Sodium Sterile Injection 1g
- Resfur RTU Ceftiofur Hydrochloride Injection 50 ml
- X Nel Ceftiofur Sodium Injection 1g
- Nutritop Multivitamin & Mineral Supplement
- Bayrocin Enorofloxcin Tablets 150 mg
- Bayrocin Enrofloxacin Tablets 50 mg
- Drontal Plus Tablet
- Bayrocin Enrofloxacin 10% Injection
- AreioCal Calcium Tablets
- AreioCal Calcium Supplement for Bones 30s Tablet
- Bayrocin 1 Shot Injection 30 ml
- Enroko 50 Enrofloxacin 50mg Tablet
- Goclin 150mg (Clindamycin) Tablet
- Goclin 300mg (Clindamycin) Tablet
- JUMP BUDDY Carprofen 100MG TABLET
- Jump Buddy (Carprofen) 50mg Tablet
- KICK TAPE CAT TABLET
- KICK TAPE DOG TABLET
- Kove Calcium Tablets
- KTD XL Praziquantel+Pyrantel +Febantel Tablet
- Petmedin 2.5mg Pimobendan Chewable Tablet
- Petmedin 5mg Pimobendan Chewable Tablet
- Vetopic Cyclosporine 50mg Tablet
- Wormstop praziquantel, pyrantel pamoate, and febantel Tablet
- Paws Up bone joint supplement tablets
- Vencard Tablet (Heart Support)
- Vendisc Tablets Joint Support
- Bandykind Plus Albendazole 3 gm + Ivermectin 100 mg Bolus
- CALCIMUST-MLD BOLUS
- NUROKIND XL MECOBALAMIN 10ml Injection
- ATOVABRUT Atovaquone 250MG TABLETS
- BANDYSTAR-CAT Praziquantel and Pyrantel Pamoate TABLETS
- Bandystar Plus praziquantel+pyrantel embonate and febantel Tablets
- Caniboon Tablets
- Cefastar-CV 200mg Tablets
- Coxkind -227 Firocoxib Chewable Tablets
- Coxkind-57 Firocoxib Chewable Tablets
- Multistar Chew Multivitamin Tablets
- Nuforce-Pet Itraconazole 100mg tablets
- CEFTABRUT-TAZO 4.5 GM Injection
- ENROSTRONG-10% Enrofloxacin 100ml INJECTION
- ENROSTRONG-PZ BOLUS
- ENROSTRONG-PZ 5gm Enrofloxacin, Pefloxacin Mesilate, and Zinc INJECTION
- SERAKIND PLUS MELOXICAM, PARACETAMOL, and SERRATIOPEPTIDASE BOLUS
- TOLFIKIND-MR Tolfenamic Acid, Paracetamol, and Chlorzoxazone Bolus
- 3-Care Halquinol Bolus
- Bovizox 3gm Ceftizoxime Sodium Injection
- Polygesic Nimesulide+Pitofenone Hydrochloride+Fenpiverinium Bromide 100ml Injection
- PYRIDASE nimesulide, paracetamol, and serratiopeptidase BOLUS
- Vetaceph 3gm Ceftriaxone sodium Injection
- Vetaceph 4gm Ceftriaxone sodium Injection
- Vetaceph Plus 4.5gm Ceftriaxone sodium and Sulbactam Injection
- Prazisam Plus Praziquantel+Pyrantel+Fenbendazole Tablets
- WOCEF-0.5 Ceftriaxone 500mg Injection
- Agrimin i Mineral Feed Supplement Bolus
- AMOXIRUM FORTE 4.5g Amoxycillin+Sulbactam Injection
- AMOXIRUM Amoxycillin Trihydrate TABLETS
- FORTIVIR 10% Enrofloxacin 30ml Injection
- FORTIVIR 10% Enrofloxacin 100ml Injection
- Gardplus 4.5g Cefoperazone Sodium+sulbactam Sodium Injection
- Hitek F Ivermectin(10.0 Mg)+Clorsulon(100.0 Mg)10ml Injection
- HITEK Ivermectin 100ml Injection
- HITEK Ivermectin 10ml Injection
- HITEK Ivermectin 30ml Injection
- HITEK PLATINUM Ivermectin 10ml Injection
- Mamiup Nutritional Feed Supplements Bolus
- MEGLUDYNE Flunixin Meglumine 100ml Injection
- MEGLUDYNE Flunixin Meglumine 20ml Injection
- Pregheat Oestradiol Benzoate 2ml Injection
- PREGOVA Cloprostenol Sodium 2ml Injection
- RUMERIC B-Complex & Amino Acids 100ml Injection
- RUMERIC B-Complex and Amino acids 30ml Injection
- AMOXIRUM FORTE Amoxycillin+Sulbactam 300mg Injection
- Canitone LS Liver Support Tablets
- CANITONE Nutritional Supplement TABLET
- FELIGEN CRP 1ml Injection
- Ipraz IVERMECTIN + PRAZIQUANTEL Tablets
- Lixen Cephalexin 300mg Tablets
- Lixen Cephalexin 600mg Tablets
- Nutrich nutritional supplement 60 Tablets
- Nutrich nutritional supplement 30 Tablets
- Zoletil 100 Injection
- ZOLETIL 50 tiletamine and zolazepam Injection
- Bovirum Vet Bolus
- Cyclomin 7 Minerals Feed Supplement Bolus
- BIOTRIM-DS Sulphadiazine+Trimethoprim BOLUS
- BIOTRIM-IM Sulphadiazine+Trimethoprim 30ml Injectionl
- Cefstan Ceftriaxone 3gm Injection
- DEXONA-VET 4.4MG DEXAMETHASONE 5ml Injection
- DICRYSTICIN-DS STREPTO-PENICILLIN 5gm Injection
- ISOFLUD ISOFLUPREDONE ACETATE 5ml Injection
- Isoflud ISOFLUPREDONE ACETATE 10ml Injection
- Isoflud ISOFLUPREDONE ACETATE 30ml Injection
- Neoprofen Ketoprofen 100mg/100ml Injection
- OXYNEX Oxytetracycline Hydrochloride 100ml Injection
- Siquil 20mg/5ml Injection
- STECLIN TETRACYCLINE BOLUS
- TissuAid Pet Feed Supplement tablet
- VETALOG TRIAMCINOLONE ACETONIDE 6mg/5ml Injecton
- M-Ceft 2500mg Ceftizoxime Injection
- M-ceft 3000mg Ceftizoxime Injection
- SURRAL Isometamidium Chloride Hcl 250mg INJECTION
- Vetcort Isoflupredone acetate 30ml Injection
- Xceft 500mg Ceftiofur sodium Injection
- Xceft 1g Ceftiofur sodium Injection
- Zeet Chlorpheniramine 100ml Injection
- Jeri Pet Capsule Joint Health Supplement
- APTIFAST Feed Supplement Bolus
- Derilan Cefquinome 500mg Injection
- Moxiwell Moxifloxacin 30ml Injection
- PROTRIO Nimesulide+Dimethyl Sulfoxide+Paracetamol 100ml Injection
- Stimmilk PTM Bolus
- Stimvet 21 Bolus
- Rabisin Rabies Vaccine 10ml Injection
- Rabisin 1ml Inactivated Rabies Antigen Injection
- Cleanex Nitrofurazone +Metronidazole +Povidone Iodine Bolus
- Mecovet-Xl 30ml Methylcobalamin Injection
- Metphos Butaphosphan+Mecobalamin 100ml Injection
- Zakshot-Plus Tranexamic Acid & Etamsylate Bolus
- Zolcol-D D-Cloprostenol 2ml Injection
- Absolute Digestive Tablets
- Absolute Immunity Tablets
- Absolute Mobility Tablet
- Viteral Plus multivitamin & multimineral supplement Tablets
- ANBIOFLAM-400 Capsule Anti-Inflammatory and Antibacterial
- Ovimin Bolus Feed Supplement
- Feritas Bolus Iron Supplement
- Feritas Iron Sorbitol 10ml Injection
- Gynarich Buserelin 5ml Injection
- Gynarich Buserelin 10ml Injection
- Gynarich Buserelin 2.5ml Injection
- Intavita-H 10ml Injection
- Intavita 10ml Injection
- NoviZac 50ml Injection
- Pragma Cloprostenol 2ml Injection
- Unizif Flunixin Meglumine 100ml Injection
- Xyrofur Ceftiofur Sodium 250mg Injection
- Xyrofur Ceftiofur Sodium 0.5gm Injection
- Xyrofur Ceftiofur Sodium 1gm Injection
- Xyrofur Tazo Ceftiofur and Tazobactam 1.125gm Injection
- Bravecto 250mg(Fluralaner)4.5kg-10kg Tablet
- Bravecto (Fluralaner) 10kg-20kg Tablets
- UTS ADVANCE TABLET
- PetClind 450mg clindamycin tablets
- PetClind 600mg clindamycin tablets
- Pet Joint Plus Joint Health Support Tablets
- Wormtrap praziquantel, pyrantel pamoate and febantel Tablet
- Megloc Advance Bolus
- Olone-CEF Plus Bolus
- Olone TZ DD Bolus
- Beetapet-250 Amoxicillin 200mg + Clavulanic Acid 50mg Tablet
- Beetapet-375 Amoxicillin 300mg + Clavulanic Acid 75mgTablet
- Carebest Ketoconazole 200 mg Tablet
- CEF - DP Cefpodoxime Proxetil 100MG TABLET
- Cef-Dp Cefpodoxime Proxetil 200mg tablet
- Deep Tbr Nutritional Supplement Tablets
- DOXY MUST 100 Doxycycline Hyclate TABLET
- DOXY MUST Doxycycline Hyclate 300mg TABLETS
- ENDRO PET 150MG Enrofloxacin Tablets
- Endro Pet 50mg Enrofloxacin Tablet
- FlexiRun hip and joint supplement Tablet
- Skyworm Cat PRAZIQUANTEL + PYRANTEL EMBONATE Tablet
- Skyworm PRAZIQUANTEL + PYRANTEL EMBONATE + FEBANTEL Tablet
- Skyworm XL PRAZIQUANTEL + PYRANTEL EMBONATE + FEBANTEL Tablet
- WELLRICH Multivitamin & Mineral TABLETS
- Vetopen Lornoxicam Paracetamol Bolus
- FLUKARID-DS Oxyclozanide, Levamisole, and Silymarin Bolus
- MEGLULAST Flunixin Meglumine 100ml Injection
- Nurokind-Plus Vet Mecobalamin 100ml Injection
- OFLOKIND-OZ Ofloxacin and Ornidazole Bolus
- OVULANTA-KIT Clomifene+Copper Sulphate Tablet
- OVULANTA-KIT Clomifene Citrate and Copper Sulphate Pentahydrate Tablet
- SERAKIND Serratiopeptidase BOLUS
- Spasmovet Dicyclomine hydrochloride 30ml Injection
- TEXABLEED Tranexamic acid 30ml Injection
- Totavit Mineral Supplement Bolus
- Totavit Mineral Supplement Bolus
- Doloban Plus Vet Bolus
- FENDIKIND-PLUS Fenbendazole and Ivermectin Bolus
- METAWAYS phosphorus & Vit. B12 30ml Injection
- MOXIKIND-CLAV 3.6 INJECTION
- Allergia Tablets
- CanWorm Praziquantel+Pyrantel pamoate+febantel Tablets
- Cardio Support Tablets
- Fecal Deterrent Tablets
- KEPHELEXIN VET Cephalexin 300mg Tablets
- Mind Calming Tablets
- Nerve On Tablets
- Uro Support Tablets
- Vetconazole Itraconazole 100mg Capsules
- VETINA GABAPENTIN 100MG TABLET
- Vetina Levetiracetam-500 mg Tablet
- Well Up tablets Nutritional Supplement
- Analgon 3000 Albendazole 3gm Bolus
- Beekom-L 100ml Injection Vitamin B complex and liver extract
- Endact LA Ivermectin 10ml Injection
- Meriquin Enrofloxacin 10% 100ml Injection
- Proxyvet MP meloxicam and paracetamol Bolus
- RIDEMA Frusemide 10ml Injection
- Rintose 500ml Injection
- Tolfine 4% 100ml Injection
- Tolfine Tolfenamic Acid Bolus
- TRIQUIN 2.5g Quinapyramine+Quinapyramine chloride Injection
- Wocef XP-3.375g ceftriaxone and Tazobactam Injection
- Cafede Praziquantel, pyrantel pamoate Tablet & febantel
- Atosporin-50 (Cyclosporin)50mg Capsules
- Atosporin-100 (Cyclosporin)100mg Capsules
- Atovaquin-250 Atovaquone 250mg tablets
- Ben-Spiro 80/10 Chewable Tablets
- Firotail 57 (Firocoxib) Tablets
- Firotail 227 (Firocoxib) Tablets
- Relaxzyme Wound Healing Tablets
- RELAXZYME M/L Tablets
- SUBAvet-IT 50 (Itraconazole) Capsules
- Terbopet 500mg (Terbinafine) Tablets
- TOCERAPET-15 Toceranib phosphate Tablets
- Vet Maro Maropitant Citrate 24mg Tablets
- CK-Reno Capsules
- CK-Reno Feline Capsules
- Cranbact Capsules
- Pancresolve Tablets
- Pimoben 1.25/2.5mg (pimobendan & benazepril) Tablet
- Pimoben 5/10 (pimobendan & benazepril) Tablets
- Pimoben-Solo 5 Pimobendan Chewable Tablets
- Rinonadyl Capsules
- Synfosium Capsules
- Ursopet Ursodeoxycholic Acid Tablets
- Vet Thyro 0.2 Levothyroxine sodium Tablets
- Vet Thyro 0.6 Levothyroxine sodium Tablets
- Aaram Ranitidine and Dimethicone Bolus
- Amcox Amoxycillin and Cloxacillin 3.5gm Injection
- Anicef-S Ceftriaxone 3gm Injection
- Best-XL Cefixime, Serratiopeptidase and Lactic acid bacillus Bolus
- Wigclav 250 AMOXYCILLIN and POTASSIUM CLAVULANATE Tablets
- Wigclav 500 AMOXYCILLIN and POTASSIUM CLAVULANATE Tablets
- Alfanil Praziquantel, Pyrantel embonate and oxantel embonate Tablets
- Nutrical-CA Tablets
- Artizone-S Phenylbutazone and Sodium Salicylate 100ml Injection
- Babimido Imidocarb Dipropionate 10ml Injection
- Belamyl 30ml Injection
- Belamyl 100ml Injection
- Clostenol Cloprostenol 2ml Injection
- Duromect Doramectin 10ml Injection
- ENRODAC- 10 Enrofloxacin 100ml Injection
- Flunimeg Flunixin Meglumine 30ml Injection
- Injectiphos Butaphosphan and Cyanocobalamin 100ml Injection
- LEMASOL-75 Levamisole 30ml Injection
- Marbodac Marbofloxacin 30ml Injection
- Neuroxin-M 10ml Injection
- Neuroxin-M 30ml Injection
- Vitamine Suppliments
- Omega3 Fatty Acid Omega-6 Fatty Acid Cod Liver Oil Tocorienols Astaxanthin And Vitamin C Tablets
- Desirox Deferasirox Tablet 250mg
- Encicarb Elemental Iron Injection 500mg
- Ferinject Ferric Carboxymaltose Injection 500mg
- Ferinject Ferric Carboxymaltose Injection 1000mg
- Injek Phytomenadione Injection 1mg
- Irem Iron Sucrose Injection 100mg
- Multiregain R Multivitamin Injection
- Nexiron Iron Sucrose Injection 100 mg
- Optineuron Forte Thiamine Vitamin B1, Vitamin B6 & D-Panthenol Injection50mg
- Optineuron Vitamin B &Vitamin B12 Injection
- Orofer Ferric Carboxymaltose Injectio 50mg
- Orofer Ferric Carboxymaltose Injection 500mg
- Orofer Ferric Carboxymaltose Injection 750 mg
- Orofer Iron Sucrose Injection 100mg
- Orofer Iron Sucrose Injection 200mg
- Nexit Drenching Uterine Tonic & Cleanser Capsules
- Ivermectin Tablets
- Urine Infective Medicine
- Finasteride Tablet
- 1 MG Finasteride Tablets IP
- Eema Hmg Menotrophin Injection 75IU
- Eugon Menotrophin Injection 75i.u
- Eugon HP Injection 150 IU
- Eutrig chorionic gonadotropin Injection 2000IU
- Eutrig HP chorionic gonadotropin Injection 10000IU
- Eutrig HP chorionic gonadotropin Injection 5000IU
- Hucog HP chorionic gonadotropin Injection 2000IU
- Hucog HP chorionic gonadotropin Injection 5000IU
- HUCOG HP chorionic gonadotropin Injection 10000IU
- Minoxidil Topical Solution
- Luliconazole Cream
- Urology Medicines
- Acetylcysteine Tablet
- Brain Nervous And Anti Depresant Medicines
- Contact Us
- Home
- Products
- Anti Cancer Medicines
- Vivitra Trastuzumab Injection 150mg
Vivitra Trastuzumab Injection 150mg
Price 9995.0 INR/ Piece
Vivitra Trastuzumab Injection 150mg Specification
- Origin
- India
- Salt Composition
- Trastuzumab
- Dosage Form
- Injection
- Indication
- Cancer treatment
- Storage Instructions
- Cool and dry place
- Shelf Life
- 12 Months
Vivitra Trastuzumab Injection 150mg Trade Information
- Minimum Order Quantity
- 1 Piece
- Supply Ability
- 100 Pieces Per Month
- Delivery Time
- 7 Days
About Vivitra Trastuzumab Injection 150mg
Site of Application & Unique Features
Vivitra Trastuzumab Injection 150mg is specifically administered via intravenous injection, making it suitable for clinical oncology care. It is used in treating certain types of cancer, offering targeted therapy for improved patient outcomes. Noteworthy for its precision and reliability, the product is formulated to meet high standards of safety and efficacy. Vivitra stands out due to its origin in India, stringent quality controls, and ease of integration into established oncology protocols.
Sample Policy & Export Credentials
Vivitra Trastuzumab Injection 150mg samples are available for quality assurance to our esteemed clients. Packaged meticulously to international standards, this product ensures secure shipping and protected freight throughout transit. The product comes with essential certifications, indicating compliance with global pharmaceutical guidelines. Our main export markets include Asia, Africa, and Europe, ensuring wide accessibility. Clients benefit from seamless logistics, efficient freight handling, and optimal packaging that maintains the medication's integrity until it reaches its final destination.
FAQ's of Vivitra Trastuzumab Injection 150mg:
Q: How is Vivitra Trastuzumab Injection 150mg administered?
A: Vivitra Trastuzumab Injection 150mg is administered intravenously by a healthcare professional, typically in a hospital or clinical setting, ensuring precise dosing and monitoring during cancer treatment.Q: What are the storage requirements for Vivitra Trastuzumab Injection 150mg?
A: This medication should be stored in a cool and dry place, away from direct sunlight and humidity, to maintain its effectiveness throughout the 12-month shelf life.Q: When should Vivitra Trastuzumab Injection 150mg be used?
A: Vivitra is prescribed for cancer patients based on a doctor's evaluation, particularly for those requiring targeted therapy involving the active ingredient Trastuzumab. It is usually given following specific clinical criteria.Q: Where is Vivitra Trastuzumab Injection 150mg manufactured and exported from?
A: Vivitra Trastuzumab Injection is produced in India by top exporters and suppliers, ensuring international quality and compliance with pharmaceutical export standards.Q: What are the benefits of using Vivitra Trastuzumab Injection 150mg?
A: The key benefits include effective cancer treatment through targeted therapy, high safety profile, notable shelf life, and availability from reputed suppliers, providing trustworthy oncology care.Q: How secure is the delivery and packaging process?
A: Vivitra is packaged following strict guidelines to ensure product integrity. Secure shipping and reliable freight partners guarantee the medication arrives safely at its destination.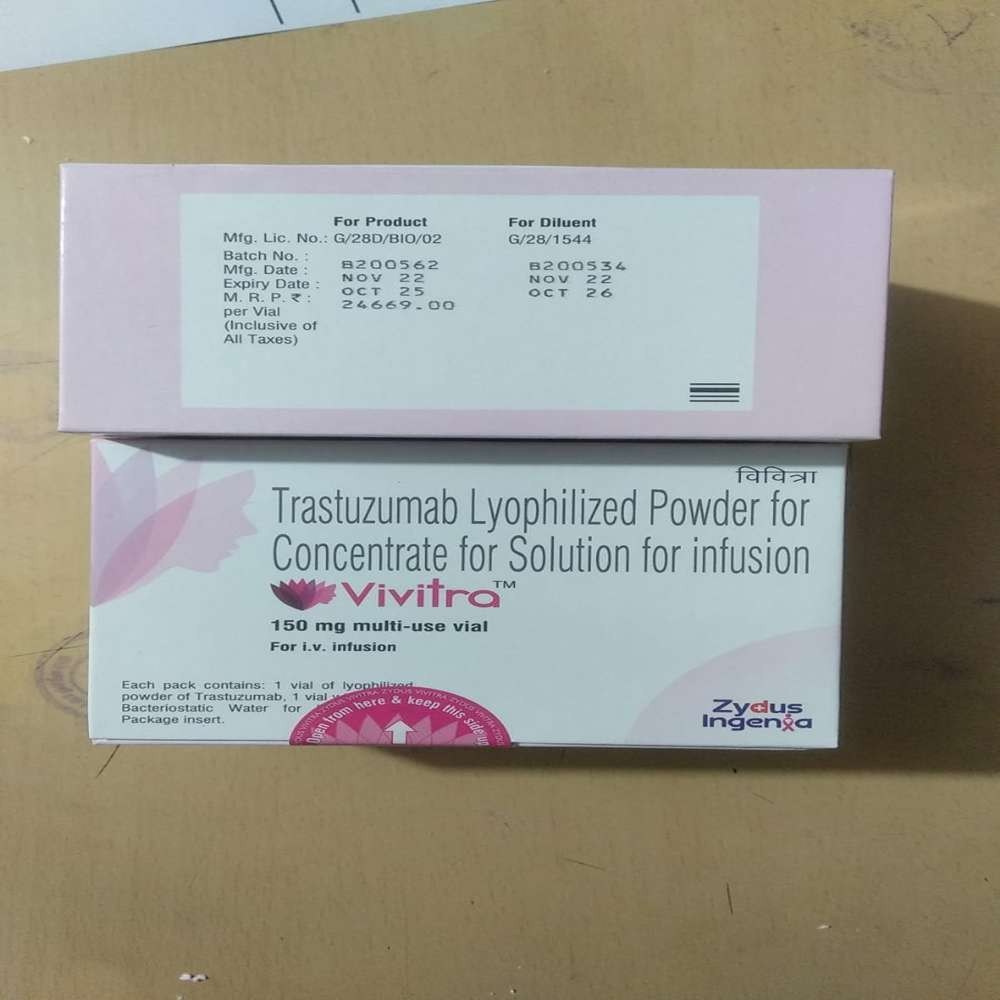



Price:
- 50
- 100
- 200
- 250
- 500
- 1000+

More Products in Anti Cancer Medicines Category
Lenangio Lenalidomide Capsule 5mg
Price 632.0 INR / Bottle
Minimum Order Quantity : 1 Bottle
Dosage Form : Capsules
Salt Composition : Lenalidomide
Origin : India
Shelf Life : 12 Months
Capegard Capecitabine Tablet 500 mg
Price 761.0 INR / Box
Minimum Order Quantity : 1 Box
Dosage Form : Tablets
Salt Composition : Capecitabine
Origin : India
Shelf Life : 12 Months
Abamune L Abacavir & Lamivudine Tablets
Price 2996.04 INR / Box
Minimum Order Quantity : 1 Box
Dosage Form : Tablets
Salt Composition : Abacavir & Lamivudine
Origin : India
Shelf Life : 12 Months
Imatib-100 Imatinib 100 Mg Tablet
Price 557.64 INR / Box
Minimum Order Quantity : 1 Box
Dosage Form : Tablets
Salt Composition : Imatinib
Origin : India
Shelf Life : 12 Months



Thank You!
Thank You for your valuable time. We have received your details and will get back to you shortly.
GST : 27AAYFK6141L1ZL
- 4th Floor, Ganjipeth Shop No 412, Central Avenue R, Sandesh Dawa Bazaar, Nagpur - 440018, Maharashtra, India
- Phone :08071931667
- Mr Siddharth (Partner)
- Mobile :08071931667
Developed and Managed by Infocom Network Private Limited.

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry